Channelopathy of small- and intermediate-conductance Ca2+-activated K+ channels
- PMID: 35715699
- PMCID: PMC9889811
- DOI: 10.1038/s41401-022-00935-1
Channelopathy of small- and intermediate-conductance Ca2+-activated K+ channels
Abstract
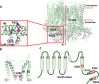
Small- and intermediate-conductance Ca2+-activated K+ (KCa2.x/KCa3.1 also called SK/IK) channels are gated exclusively by intracellular Ca2+. The Ca2+ binding protein calmodulin confers sub-micromolar Ca2+ sensitivity to the channel-calmodulin complex. The calmodulin C-lobe is constitutively associated with the proximal C-terminus of the channel. Interactions between calmodulin N-lobe and the channel S4-S5 linker are Ca2+-dependent, which subsequently trigger conformational changes in the channel pore and open the gate. KCNN genes encode four subtypes, including KCNN1 for KCa2.1 (SK1), KCNN2 for KCa2.2 (SK2), KCNN3 for KCa2.3 (SK3), and KCNN4 for KCa3.1 (IK). The three KCa2.x channel subtypes are expressed in the central nervous system and the heart. The KCa3.1 subtype is expressed in the erythrocytes and the lymphocytes, among other peripheral tissues. The impact of dysfunctional KCa2.x/KCa3.1 channels on human health has not been well documented. Human loss-of-function KCa2.2 mutations have been linked with neurodevelopmental disorders. Human gain-of-function mutations that increase the apparent Ca2+ sensitivity of KCa2.3 and KCa3.1 channels have been associated with Zimmermann-Laband syndrome and hereditary xerocytosis, respectively. This review article discusses the physiological significance of KCa2.x/KCa3.1 channels, the pathophysiology of the diseases linked with KCa2.x/KCa3.1 mutations, the structure-function relationship of the mutant KCa2.x/KCa3.1 channels, and potential pharmacological therapeutics for the KCa2.x/KCa3.1 channelopathy.
Keywords: KCa2.2 channels; KCa2.3 channels; KCa3.1 channels; Zimmermann-Laband syndrome; channelopathy; hereditary xerocytosis.
© 2022. The Author(s), under exclusive licence to Shanghai Institute of Materia Medica, Chinese Academy of Sciences and Chinese Pharmacological Society.
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Channelopathy-causing mutations in the S45A/S45B and HA/HB helices of KCa2.3 and KCa3.1 channels alter their apparent Ca2+ sensitivity.Cell Calcium. 2022 Mar;102:102538. doi: 10.1016/j.ceca.2022.102538. Epub 2022 Jan 8. Cell Calcium. 2022. PMID: 35030515 Free PMC article.
-
Laminar shear stress upregulates endothelial Ca²⁺-activated K⁺ channels KCa2.3 and KCa3.1 via a Ca²⁺/calmodulin-dependent protein kinase kinase/Akt/p300 cascade.Am J Physiol Heart Circ Physiol. 2013 Aug 15;305(4):H484-93. doi: 10.1152/ajpheart.00642.2012. Epub 2013 Jun 21. Am J Physiol Heart Circ Physiol. 2013. PMID: 23792675 Free PMC article.
-
Pharmacology of Small- and Intermediate-Conductance Calcium-Activated Potassium Channels.Annu Rev Pharmacol Toxicol. 2020 Jan 6;60:219-240. doi: 10.1146/annurev-pharmtox-010919-023420. Epub 2019 Jul 23. Annu Rev Pharmacol Toxicol. 2020. PMID: 31337271 Free PMC article. Review.
-
Novel phenolic inhibitors of small/intermediate-conductance Ca²⁺-activated K⁺ channels, KCa3.1 and KCa2.3.PLoS One. 2013;8(3):e58614. doi: 10.1371/journal.pone.0058614. Epub 2013 Mar 14. PLoS One. 2013. PMID: 23516517 Free PMC article.
-
Pharmacological gating modulation of small- and intermediate-conductance Ca(2+)-activated K(+) channels (KCa2.x and KCa3.1).Channels (Austin). 2015;9(6):336-43. doi: 10.1080/19336950.2015.1071748. Epub 2015 Jul 28. Channels (Austin). 2015. PMID: 26217968 Free PMC article. Review.
Cited by
-
KCa 2.2 (KCNN2): A physiologically and therapeutically important potassium channel.J Neurosci Res. 2023 Nov;101(11):1699-1710. doi: 10.1002/jnr.25233. Epub 2023 Jul 19. J Neurosci Res. 2023. PMID: 37466411 Free PMC article. Review.
-
ACYP2 functions as an innovative nano-therapeutic target to impede the progression of hepatocellular carcinoma by inhibiting the activity of TERT and the KCNN4/ERK pathway.J Nanobiotechnology. 2024 Sep 12;22(1):557. doi: 10.1186/s12951-024-02827-4. J Nanobiotechnology. 2024. PMID: 39267048 Free PMC article.
-
A sexually dimorphic signature of activity-dependent BDNF signaling on the intrinsic excitability of pyramidal neurons in the prefrontal cortex.Front Cell Neurosci. 2024 Nov 6;18:1496930. doi: 10.3389/fncel.2024.1496930. eCollection 2024. Front Cell Neurosci. 2024. PMID: 39569070 Free PMC article.
-
Simulations predict differing phase responses to excitation vs. inhibition in theta-resonant pyramidal neurons.J Neurophysiol. 2023 Oct 1;130(4):910-924. doi: 10.1152/jn.00160.2023. Epub 2023 Aug 23. J Neurophysiol. 2023. PMID: 37609720 Free PMC article.
-
hiPSC-derived cardiomyocytes as a model to study the role of small-conductance Ca2+-activated K+ (SK) ion channel variants associated with atrial fibrillation.Front Cell Dev Biol. 2024 Jan 18;12:1298007. doi: 10.3389/fcell.2024.1298007. eCollection 2024. Front Cell Dev Biol. 2024. PMID: 38304423 Free PMC article. Review.
References
-
- Aldrich RW, Chandy KG, Grissmer S, Gutman GA, Kaczmarek LK, Wei AD, et al. Calcium- and sodium-activated potassium channels (version 2019.4) in the IUPHAR/BPS Guide to Pharmacology Database. IUPHAR/BPS Guide to Pharmacology CITE. 2019.
-
- Higham J, Sahu G, Wazen RM, Colarusso P, Gregorie A, Harvey BSJ, et al. Preferred formation of heteromeric channels between coexpressed SK1 and IKCa Channel subunits provides a unique pharmacological profile of Ca2+-activated potassium channels. Mol Pharmacol. 2019;96:115–26. doi: 10.1124/mol.118.115634. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

