mRNA Vaccines: The Dawn of a New Era of Cancer Immunotherapy
- PMID: 35720301
- PMCID: PMC9201022
- DOI: 10.3389/fimmu.2022.887125
mRNA Vaccines: The Dawn of a New Era of Cancer Immunotherapy
Abstract
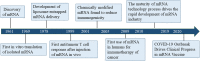
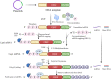
mRNA therapy is a novel anticancer strategy based on in vitro transcription (IVT), which has potential for the treatment of malignant tumors. The outbreak of the COVID-19 pandemic in the early 21st century has promoted the application of mRNA technologies in SARS-CoV-2 vaccines, and there has been a great deal of interest in the research and development of mRNA cancer vaccines. There has been progress in a number of key technologies, including mRNA production strategies, delivery systems, antitumor immune strategies, etc. These technologies have accelerated the progress and clinical applications of mRNA therapy, overcoming problems encountered in the past, such as instability, inefficient delivery, and weak immunogenicity of mRNA vaccines. This review provides a detailed overview of the production, delivery systems, immunological mechanisms, and antitumor immune response strategies for mRNA cancer vaccines. We list some mRNA cancer vaccines that are candidates for cancer treatment and discuss clinical trials in the field of tumor immunotherapy. In addition, we discuss the immunological mechanism of action by which mRNA vaccines destroy tumors as well as challenges and prospects for the future.
Keywords: clinical trials; delivery system; immunological mechanism; mRNA vaccine; malignant tumor.
Copyright © 2022 Deng, Tian, Song, An and Yang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

