Mitigating the risk of transfusion-transmitted infections with vector-borne agents solely by means of pathogen reduction
- PMID: 35726756
- PMCID: PMC9541364
- DOI: 10.1111/trf.16950
Mitigating the risk of transfusion-transmitted infections with vector-borne agents solely by means of pathogen reduction
Abstract
Background: This study evaluated whether pathogen reduction technology (PRT) in plasma and platelets using amotosalen/ultraviolet A light (A/UVA) or in red blood cells using amustaline/glutathione (S-303/GSH) may be used as the sole mitigation strategy preventing transfusion-transmitted West Nile (WNV), dengue (DENV), Zika (ZIKV), and chikungunya (CHIKV) viral, and Babesia microti, Trypanosoma cruzi, and Plasmodium parasitic infections.
Methods: Antibody (Ab) status and pathogen loads (copies/mL) were obtained for donations from US blood donors testing nucleic acid (NAT)-positive for WNV, DENV, ZIKV, CHIKV, and B. microti. Infectivity titers derived from pathogen loads were compared to published PRT log10 reduction factors (LRF); LRFs were also reviewed for Plasmodium and T. cruzi. The potential positive impact on donor retention following removal of deferrals from required questioning and testing for WNV, Babesia, Plasmodium, and T. cruzi was estimated for American Red Cross (ARC) donors.
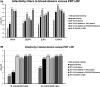
Results: A/UVA and S-303/GSH reduced infectivity to levels in accordance with those recognized by FDA as suitable to replace testing for all agents evaluated. If PRT replaced deferrals resulting from health history questions and/or NAT for WNV, Babesia, Plasmodium, and T. cruzi, 27,758 ARC donors could be retained allowing approximately 50,000 additional donations/year based on 1.79 donations/donor for calendar year 2019 (extrapolated to an estimated 125,000 additional donations nationally).
Conclusion: Pathogen loads in donations from US blood donors demonstrated that robust PRT may provide an opportunity to replace deferrals associated with donor questioning and NAT for vector-borne agents allowing for significant donor retention and likely increased blood availability.
© 2022 The Authors. Transfusion published by Wiley Periodicals LLC on behalf of AABB.
Conflict of interest statement
Marion C. Lanteri is an employee of Cerus Corporation. The remaining authors declare no conflicts of interest.
Figures

References
-
- INTERCEPT® blood system for plasma package insert, Concord, CA: Cerus Corporation; 2020.
-
- INTERCEPT® blood system for platelets package insert, Concord, CA: Cerus Corporation; 2019.
-
- U.S Department of Health and Human Services, Food and Drug Administration, Center for Biologics Evaluation and Research . Bacterial risk control strategies for blood collection establishments and transfusion services to enhance the safety and availability of platelets for transfusion ‐ guidance for industry. 2020.
-
- U.S Department of Health and Human Services, Food and Drug Administration, Center for Biologics Evaluation and Research . Recommendations for donor screening, deferral, and product management to reduce the risk of transfusion transmission of Zika virus ‐ guidance for industry. 2016. http://www.fda.gov/downloads/BiologicsBloodVaccines/GuidanceComplianceRe....
-
- U.S Department of Health and Human Services, Food and Drug Administration, Center for Biologics Evaluation and Research . Recommendations for reducing the risk of transfusion‐transmitted Babesiosis ‐ guidance for industry. 2019. https://www.fda.gov/BiologicsBloodVaccines/GuidanceComplianceRegulatoryI....
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

