The pharmacokinetic, safety, and tolerability profiles of eslicarbazepine acetate are comparable between Korean and White subjects
- PMID: 35727711
- PMCID: PMC9468563
- DOI: 10.1111/cts.13344
The pharmacokinetic, safety, and tolerability profiles of eslicarbazepine acetate are comparable between Korean and White subjects
Abstract
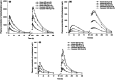
Eslicarbazepine acetate (ESL) is a prodrug antiseizure medication for the treatment of focal seizures. ESL shows a well-established pharmacokinetic (PK)-pharmacodynamic relationship and has similar extrinsic epilepsy-related factors across ethnicities. This study evaluated and compared ESL safety, tolerability, and PK characteristics between Korean and White subjects. A randomized, double-blind, placebo-controlled, single- and multiple-dose escalation study was conducted in healthy Korean and White adults. Participants randomly received a single dose and multiple oral doses of ESL (400-1600 mg) or placebo once daily for 11 days at a ratio of 8:2. Serial blood samples were collected to determine the plasma concentration of ESL and its metabolites (eslicarbazepine, [R-licarbazepine and oxcarbazepine). Safety and tolerability were assessed throughout the study. A total of 29 Korean and 20 White subjects completed the study. The PK profiles of the metabolites of ESL were similar between Korean and White subjects. The geometric mean ratio (90% confidence interval) of Korean to White subjects for the area under the concentration-time curve within a dosing interval of eslicarbazepine was 1.06 (0.97-1.17) and 0.96 (0.87-1.06) after multiple oral doses of 400 and 1600 mg ESL, respectively. Other PK parameters were also similar between the two ethnic groups. ESL was well-tolerated in healthy Korean and White subjects, and its PK characteristics were similar between the two ethnic groups. The results of this study support to use the same dosage regimen of ESL in both White and Korean patients with seizures.
© 2022 The Authors. Clinical and Translational Science published by Wiley Periodicals LLC on behalf of American Society for Clinical Pharmacology and Therapeutics.
Conflict of interest statement
The authors declared no competing interests for this work.
Figures

Similar articles
-
Pharmacokinetics of eslicarbazepine acetate at steady-state in adults with partial-onset seizures.Epilepsy Res. 2011 Sep;96(1-2):132-9. doi: 10.1016/j.eplepsyres.2011.05.013. Epub 2011 Jun 15. Epilepsy Res. 2011. PMID: 21680153 Clinical Trial.
-
Pharmacokinetics and tolerability of eslicarbazepine acetate and oxcarbazepine at steady state in healthy volunteers.Epilepsia. 2013 Aug;54(8):1453-61. doi: 10.1111/epi.12242. Epub 2013 Jun 12. Epilepsia. 2013. PMID: 23758485 Clinical Trial.
-
Pooled efficacy and safety of eslicarbazepine acetate as add-on treatment in patients with focal-onset seizures: Data from four double-blind placebo-controlled pivotal phase III clinical studies.CNS Neurosci Ther. 2017 Dec;23(12):961-972. doi: 10.1111/cns.12765. Epub 2017 Oct 13. CNS Neurosci Ther. 2017. PMID: 29030894 Free PMC article.
-
Pharmacokinetics and drug interactions of eslicarbazepine acetate.Epilepsia. 2012 Jun;53(6):935-46. doi: 10.1111/j.1528-1167.2012.03519.x. Epub 2012 May 21. Epilepsia. 2012. PMID: 22612290 Review.
-
Eslicarbazepine acetate: its effectiveness as adjunctive therapy in clinical trials and open studies.J Neurol. 2017 Mar;264(3):421-431. doi: 10.1007/s00415-016-8338-2. Epub 2017 Jan 18. J Neurol. 2017. PMID: 28101651 Free PMC article. Review.
Cited by
-
Long-term effects of adjunctive eslicarbazepine acetate in adult Asian patients with refractory focal seizures: Post hoc analysis of a phase III trial.Clin Transl Sci. 2024 May;17(5):e13802. doi: 10.1111/cts.13802. Clin Transl Sci. 2024. PMID: 38787305 Free PMC article. Clinical Trial.
-
A real-world pharmacovigilance analysis of eslicarbazepine acetate using the FDA adverse events reporting system (FAERS) database from 2013 (Q4) to 2024 (Q1).Front Pharmacol. 2024 Sep 20;15:1463560. doi: 10.3389/fphar.2024.1463560. eCollection 2024. Front Pharmacol. 2024. PMID: 39372199 Free PMC article.
-
Determination of Antiepileptics in Biological Samples-A Review.Molecules. 2024 Oct 2;29(19):4679. doi: 10.3390/molecules29194679. Molecules. 2024. PMID: 39407608 Free PMC article. Review.
-
Pharmacokinetic drug-drug interactions of JBPOS0101 mediated by cytochrome P450 3A4 and UDP-glucuronosyltransferases.Clin Transl Sci. 2024 Jul;17(7):e13892. doi: 10.1111/cts.13892. Clin Transl Sci. 2024. PMID: 39034448 Free PMC article.
References
-
- Trinka E, Ben‐Menachem E, Kowacs PA, et al. Efficacy and safety of eslicarbazepine acetate versus controlled‐release carbamazepine monotherapy in newly diagnosed epilepsy: a phase III double‐blind, randomized, parallel‐group, multicenter study. Epilepsia. 2018;59(2):479‐491. - PubMed
-
- Hainzl D, Parada A, Soares‐da‐Silva P. Metabolism of two new antiepileptic drugs and their principal metabolites S(+)‐ and R(−)‐10,11‐dihydro‐10‐hydroxy carbamazepine. Epilepsy Res. 2001;44(2–3):197‐206. - PubMed
-
- Bialer M, Soares‐da‐Silva P. Pharmacokinetics and drug interactions of eslicarbazepine acetate. Epilepsia. 2012;53(6):935‐946. - PubMed
-
- Elger C, Bialer M, Falcão A, et al. Pharmacokinetics and tolerability of eslicarbazepine acetate and oxcarbazepine at steady state in healthy volunteers. Epilepsia. 2013;54(8):1453‐1461. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

