Parcellation-based tractographic modeling of the salience network through meta-analysis
- PMID: 35733239
- PMCID: PMC9304834
- DOI: 10.1002/brb3.2646
Parcellation-based tractographic modeling of the salience network through meta-analysis
Abstract
Background: The salience network (SN) is a transitory mediator between active and passive states of mind. Multiple cortical areas, including the opercular, insular, and cingulate cortices have been linked in this processing, though knowledge of network connectivity has been devoid of structural specificity.
Objective: The current study sought to create an anatomically specific connectivity model of the neural substrates involved in the salience network.
Methods: A literature search of PubMed and BrainMap Sleuth was conducted for resting-state and task-based fMRI studies relevant to the salience network according to PRISMA guidelines. Publicly available meta-analytic software was utilized to extract relevant fMRI data for the creation of an activation likelihood estimation (ALE) map and relevant parcellations from the human connectome project overlapping with the ALE data were identified for inclusion in our SN model. DSI-based fiber tractography was then performed on publicaly available data from healthy subjects to determine the structural connections between cortical parcellations comprising the network.
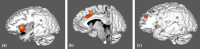
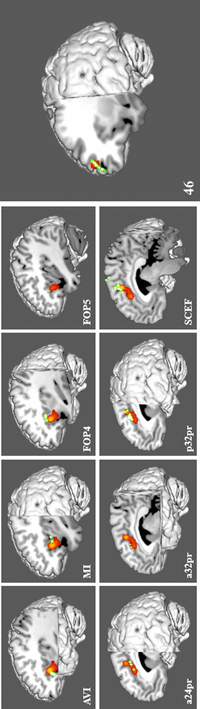
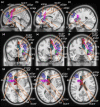
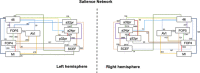
Results: Nine cortical regions were found to comprise the salience network: areas AVI (anterior ventral insula), MI (middle insula), FOP4 (frontal operculum 4), FOP5 (frontal operculum 5), a24pr (anterior 24 prime), a32pr (anterior 32 prime), p32pr (posterior 32 prime), and SCEF (supplementary and cingulate eye field), and 46. The frontal aslant tract was found to connect the opercular-insular cluster to the middle cingulate clusters of the network, while mostly short U-fibers connected adjacent nodes of the network.
Conclusion: Here we provide an anatomically specific connectivity model of the neural substrates involved in the salience network. These results may serve as an empiric basis for clinical translation in this region and for future study which seeks to expand our understanding of how specific neural substrates are involved in salience processing and guide subsequent human behavior.
Keywords: anatomy; parcellation; salience network; tractography.
© 2022 The Authors. Brain and Behavior published by Wiley Periodicals LLC.
Conflict of interest statement
Michael Sughrue is the chief medical officer, co‐founder, and shareholder of Omniscient Neurotechnology. Charles Teo is also a consultant for Aesculap and Omniscient Neurotechnology. Isabella Young is an employee of Omniscient Neurotechnology. No products related to this were discussed in this paper. No other authors report any conflict of interest.
Figures
References
-
- CJNeuroLab . (2016). HCP‐MMP1.0 volumetric (NIfTI) masks in native structural space . https://cjneurolab.org/2016/11/22/hcp‐mmp1‐0‐volumetric‐nifti‐masks‐in‐n...
-
- Baker, C. M. , Burks, J. D. , Briggs, R. G. , Conner, A. K. , Glenn, C. A. , Morgan, J. P. , Stafford, J. , Sali, G. , McCoy, T. M. , Battiste, J. D. , O'Donoghue, D. L. , & Sughrue, M. E. (2018). A connectomic atlas of the human cerebrum–Chapter 2: The lateral frontal lobe. Operative Neurosurgery (Hagerstown, MD), 15(1), S10–S74. 10.1093/ons/opy254 - DOI - PMC - PubMed
-
- Baker, C. M. , Burks, J. D. , Briggs, R. G. , Conner, A. K. , Glenn, C. A. , Robbins, J. M. , Sheets, J. R. , Sali, G. , McCoy, T. M. , Battiste, J. D. , O'Donoghue, D. L. , & Sughrue, M. E. (2018). A connectomic atlas of the human cerebrum–Chapter 5: The insula and opercular cortex. Operative Neurosurgery (Hagerstown, MD), 15(1), S175–S244. 10.1093/ons/opy259 - DOI - PMC - PubMed
-
- Baker, C. M. , Burks, J. D. , Briggs, R. G. , Conner, A. K. , Glenn, C. A. , Sali, G. , McCoy, T. M. , Battiste, J. D. , O'Donoghue, D. L. , & Sughrue, M. E. (2018). A connectomic atlas of the human cerebrum–Chapter 1: Introduction, methods, and significance. Operative Neurosurgery (Hagerstown, MD), 15(1), S1–S9. 10.1093/ons/opy253 - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

