Antibody and Memory B-Cell Immunity in a Heterogeneously SARS-CoV-2-Infected and -Vaccinated Population
- PMID: 35735743
- PMCID: PMC9426429
- DOI: 10.1128/mbio.00840-22
Antibody and Memory B-Cell Immunity in a Heterogeneously SARS-CoV-2-Infected and -Vaccinated Population
Abstract
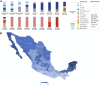
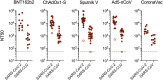
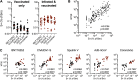
Global population immunity to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is accumulating through heterogeneous combinations of infection and vaccination. Vaccine distribution in low- and middle-income countries has been variable and reliant on diverse vaccine platforms. We studied B-cell immunity in Mexico, a middle-income country where five different vaccines have been deployed to populations with high SARS-CoV-2 incidences. Levels of antibodies that bound a stabilized prefusion spike trimer, neutralizing antibody titers, and memory B-cell expansion correlated with each other across vaccine platforms. Nevertheless, the vaccines elicited variable levels of B-cell immunity, and the majority of recipients had undetectable neutralizing activity against the recently emergent omicron variant. SARS-CoV-2 infection, experienced before or after vaccination, potentiated B-cell immune responses and enabled the generation of neutralizing activity against omicron and SARS-CoV for all vaccines in nearly all individuals. These findings suggest that broad population immunity to SARS-CoV-2 will eventually be achieved but by heterogeneous paths. IMPORTANCE The majority of studies on SARS-CoV-2 vaccine-elicited immunity and immune evasion have focused on single vaccines corresponding to those distributed in high-income countries. However, in low- and middle-income countries, vaccine deployment has been far less uniform. It is therefore important to determine the levels of immunity elicited by vaccines that have been deployed globally. Such data should help inform policy. Thus, this paper is very much a "real-world" study that focuses on a middle-income country, Mexico, in which five different vaccines based on mRNA, adenovirus, and inactivated-virus platforms have been extensively deployed, while (as documented in our study) SARS-CoV-2 variants with increasing degrees of immune evasiveness have propagated in the Mexican population, culminating in the recent emergence of B.1.1.529 (omicron).
Keywords: SARS-CoV-2; infection; memory B cells; neutralizing antibodies; vaccination.
Conflict of interest statement
The authors declare a conflict of interest. PDB has received compensation from Pfizer for consultation in the areaof SARS-CoV-2 vaccines. TH has received funding from Bristol Myers Squibb for evaluating monoclonal antibodies against SARS-CoV-2.
Figures



Update of
-
Antibody and memory B-cell immunity in a heterogeneously SARS-CoV-2 infected and vaccinated population.medRxiv [Preprint]. 2022 Feb 8:2022.02.07.22270626. doi: 10.1101/2022.02.07.22270626. medRxiv. 2022. Update in: mBio. 2022 Aug 30;13(4):e0084022. doi: 10.1128/mbio.00840-22. PMID: 35169812 Free PMC article. Updated. Preprint.
References
-
- Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, Perez JL, Pérez Marc G, Moreira ED, Zerbini C, Bailey R, Swanson KA, Roychoudhury S, Koury K, Li P, Kalina WV, Cooper D, Frenck RW, Jr, Hammitt LL, Türeci Ö, Nell H, Schaefer A, Ünal S, Tresnan DB, Mather S, Dormitzer PR, Şahin U, Jansen KU, Gruber WC, C4591001 Clinical Trial Group . 2020. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med 383:2603–2615. doi:10.1056/NEJMoa2034577. - DOI - PMC - PubMed
-
- Voysey M, Costa Clemens SA, Madhi SA, Weckx LY, Folegatti PM, Aley PK, Angus B, Baillie VL, Barnabas SL, Bhorat QE, Bibi S, Briner C, Cicconi P, Clutterbuck EA, Collins AM, Cutland CL, Darton TC, Dheda K, Dold C, Duncan CJA, Emary KRW, Ewer KJ, Flaxman A, Fairlie L, Faust SN, Feng S, Ferreira DM, Finn A, Galiza E, Goodman AL, Green CM, Green CA, Greenland M, Hill C, Hill HC, Hirsch I, Izu A, Jenkin D, Joe CCD, Kerridge S, Koen A, Kwatra G, Lazarus R, Libri V, Lillie PJ, Marchevsky NG, Marshall RP, Mendes AVA, Milan EP, Minassian AM, et al. . 2021. Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: a pooled analysis of four randomised trials. Lancet 397:881–891. doi:10.1016/S0140-6736(21)00432-3. - DOI - PMC - PubMed
-
- Logunov DY, Dolzhikova IV, Zubkova OV, Tukhvatullin AI, Shcheblyakov DV, Dzharullaeva AS, Grousova DM, Erokhova AS, Kovyrshina AV, Botikov AG, Izhaeva FM, Popova O, Ozharovskaya TA, Esmagambetov IB, Favorskaya IA, Zrelkin DI, Voronina DV, Shcherbinin DN, Semikhin AS, Simakova YV, Tokarskaya EA, Lubenets NL, Egorova DA, Shmarov MM, Nikitenko NA, Morozova LF, Smolyarchuk EA, Kryukov EV, Babira VF, Borisevich SV, Naroditsky BS, Gintsburg AL. 2020. Safety and immunogenicity of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine in two formulations: two open, non-randomised phase 1/2 studies from Russia. Lancet 396:887–897. doi:10.1016/S0140-6736(20)31866-3. - DOI - PMC - PubMed
-
- Zhu F-C, Guan X-H, Li Y-H, Huang J-Y, Jiang T, Hou L-H, Li J-X, Yang B-F, Wang L, Wang W-J, Wu S-P, Wang Z, Wu X-H, Xu J-J, Zhang Z, Jia S-Y, Wang B-S, Hu Y, Liu J-J, Zhang J, Qian X-A, Li Q, Pan H-X, Jiang H-D, Deng P, Gou J-B, Wang X-W, Wang X-H, Chen W. 2020. Immunogenicity and safety of a recombinant adenovirus type-5-vectored COVID-19 vaccine in healthy adults aged 18 years or older: a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 396:479–488. doi:10.1016/S0140-6736(20)31605-6. - DOI - PMC - PubMed
-
- Jara A, Undurraga EA, González C, Paredes F, Fontecilla T, Jara G, Pizarro A, Acevedo J, Leo K, Leon F, Sans C, Leighton P, Suárez P, García-Escorza H, Araos R. 2021. Effectiveness of an inactivated SARS-CoV-2 vaccine in Chile. N Engl J Med 385:875–884. doi:10.1056/NEJMoa2107715. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

