Host-Pathogen Interactions between Metarhizium spp. and Locusts
- PMID: 35736085
- PMCID: PMC9224550
- DOI: 10.3390/jof8060602
Host-Pathogen Interactions between Metarhizium spp. and Locusts
Abstract
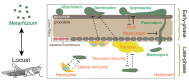
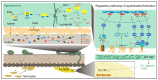
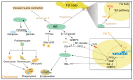
The progress in research on the interactions between Metarhizium spp. and locusts has improved our understanding of the interactions between fungal infection and host immunity. A general network of immune responses has been constructed, and the pathways regulating fungal pathogenicity have also been explored in depth. However, there have been no systematic surveys of interaction between Metarhizium spp. and locusts. The pathogenesis of Metarhizium comprises conidial attachment, germination, appressorial formation, and colonization in the body cavity of the host locusts. Meanwhile, the locust resists fungal infection through humoral and cellular immunity. Here, we summarize the crucial pathways that regulate the pathogenesis of Metarhizium and host immune defense. Conidial hydrophobicity is mainly affected by the contents of hydrophobins and chitin. Appressorial formation is regulated by the pathways of MAPKs, cAMP/PKA, and Ca2+/calmodulin. Lipid droplets degradation and secreted enzymes contributed to fungal penetration. The humoral response of locust is coordinated by the Toll pathway and the ecdysone. The regulatory mechanism of hemocyte differentiation and migration is elusive. In addition, behavioral fever and density-dependent population immunity have an impact on the resistance of hosts against fungal infection. This review depicts a prospect to help us understand host-pathogen interactions and provides a foundation for the engineering of entomopathogenic fungi and the discovery of insecticidal targets to control insect pests.
Keywords: Metarhizium; immune response; locust; pathogenicity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Vega F.E., Meyling N.V., Luangsa-Ard J.J., Blackwell M. Fungal Entomopathogens. In: Vega F., Kaya H., editors. Insect Pathology. 2nd ed. Academic; San Diego, CA, USA: 2012. pp. 171–220.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

