Reagent-Free Immobilization of Industrial Lipases to Develop Lipolytic Membranes with Self-Cleaning Surfaces
- PMID: 35736306
- PMCID: PMC9229154
- DOI: 10.3390/membranes12060599
Reagent-Free Immobilization of Industrial Lipases to Develop Lipolytic Membranes with Self-Cleaning Surfaces
Abstract
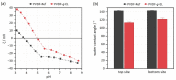
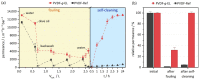
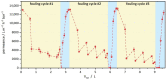
Biocatalytic membrane reactors combine the highly efficient biotransformation capability of enzymes with the selective filtration performance of membrane filters. Common strategies to immobilize enzymes on polymeric membranes are based on chemical coupling reactions. Still, they are associated with drawbacks such as long reaction times, high costs, and the use of potentially toxic or hazardous reagents. In this study, a reagent-free immobilization method based on electron beam irradiation was investigated, which allows much faster, cleaner, and cheaper fabrication of enzyme membrane reactors. Two industrial lipase enzymes were coupled onto a polyvinylidene fluoride (PVDF) flat sheet membrane to create self-cleaning surfaces. The response surface methodology (RSM) in the design-of-experiments approach was applied to investigate the effects of three numerical factors on enzyme activity, yielding a maximum activity of 823 ± 118 U m-2 (enzyme concentration: 8.4 g L-1, impregnation time: 5 min, irradiation dose: 80 kGy). The lipolytic membranes were used in fouling tests with olive oil (1 g L-1 in 2 mM sodium dodecyl sulfate), resulting in 100% regeneration of filtration performance after 3 h of self-cleaning in an aqueous buffer (pH 8, 37 °C). Reusability with three consecutive cycles demonstrates regeneration of 95%. Comprehensive membrane characterization was performed by determining enzyme kinetic parameters, permeance monitoring, X-ray photoelectron spectroscopy, FTIR spectroscopy, scanning electron microscopy, and zeta potential, as well as water contact angle measurements.
Keywords: electron beam; enzyme membrane reactor; fouling; lipase; response surface methodology; self-cleaning surface.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures



Similar articles
-
Highly Efficient One-Step Protein Immobilization on Polymer Membranes Supported by Response Surface Methodology.Front Chem. 2022 Jan 18;9:804698. doi: 10.3389/fchem.2021.804698. eCollection 2021. Front Chem. 2022. PMID: 35118049 Free PMC article.
-
Multienzyme Immobilization on PVDF Membrane via One-Step Mussel-Inspired Method: Enhancing Fouling Resistance and Self-Cleaning Efficiency.Membranes (Basel). 2024 Sep 27;14(10):208. doi: 10.3390/membranes14100208. Membranes (Basel). 2024. PMID: 39452819 Free PMC article.
-
Bio-Inspired Polymer Membrane Surface Cleaning.Polymers (Basel). 2017 Mar 9;9(3):97. doi: 10.3390/polym9030097. Polymers (Basel). 2017. PMID: 30970776 Free PMC article.
-
Fabrication of photo-Fenton self-cleaning PVDF composite membrane for highly efficient oil-in-water emulsion separation.RSC Adv. 2022 Dec 12;12(55):35543-35555. doi: 10.1039/d2ra07116a. eCollection 2022 Dec 12. RSC Adv. 2022. PMID: 36540403 Free PMC article.
-
Surface-Engineered Biocatalytic Composite Membranes for Reduced Protein Fouling and Self-Cleaning.ACS Appl Mater Interfaces. 2018 Aug 15;10(32):27477-27487. doi: 10.1021/acsami.8b07945. Epub 2018 Aug 6. ACS Appl Mater Interfaces. 2018. PMID: 30048587
Cited by
-
Bibliometrics of Functional Polymeric Biomaterials with Bioactive Properties Prepared by Radiation-Induced Graft Copolymerisation: A Review.Polymers (Basel). 2022 Nov 10;14(22):4831. doi: 10.3390/polym14224831. Polymers (Basel). 2022. PMID: 36432958 Free PMC article. Review.
-
Pyrazine derivative synthesis in a continuous-flow system: a green synthesis of pyrazinamide from pyrazine esters and amines catalyzed by Lipozyme® TL IM from Thermomyces lanuginosus.RSC Adv. 2024 Dec 16;14(53):39560-39568. doi: 10.1039/d4ra06761d. eCollection 2024 Dec 10. RSC Adv. 2024. PMID: 39687336 Free PMC article.
-
Production and Bioconversion Efficiency of Enzyme Membrane Bioreactors in the Synthesis of Valuable Products.Membranes (Basel). 2023 Jul 16;13(7):673. doi: 10.3390/membranes13070673. Membranes (Basel). 2023. PMID: 37505039 Free PMC article. Review.
References
-
- Prazeres D.M.F., Cabral J.M.S. Enzymatic membrane bioreactors and their applications. Enzym. Microb. Technol. 1994;16:738–750. doi: 10.1016/0141-0229(94)90030-2. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

