HO-1 Limits the Efficacy of Vemurafenib/PLX4032 in BRAFV600E Mutated Melanoma Cells Adapted to Physiological Normoxia or Hypoxia
- PMID: 35740068
- PMCID: PMC9219655
- DOI: 10.3390/antiox11061171
HO-1 Limits the Efficacy of Vemurafenib/PLX4032 in BRAFV600E Mutated Melanoma Cells Adapted to Physiological Normoxia or Hypoxia
Abstract
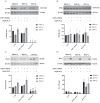
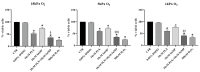
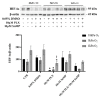
Induction of heme oxygenase 1 (HO-1) favors immune-escape in BRAFV600 melanoma cells treated with Vemurafenib/PLX4032 under standard cell culture conditions. However, the oxygen tension under standard culture conditions (~18 kPa O2) is significantly higher than the physiological oxygen levels encountered in vivo. In addition, cancer cells in vivo are often modified by hypoxia. In this study, MeOV-1 primary melanoma cells bearing the BRAFV600E mutation, were adapted to either 5 kPa O2 (physiological normoxia) or 1 kPa O2 (hypoxia) and then exposed to 10 μM PLX4032. PLX4032 abolished ERK phosphorylation, reduced Bach1 expression and increased HO-1 levels independent of pericellular O2 tension. Moreover, cell viability was significantly reduced further in cells exposed to PLX4032 plus Tin mesoporphyrin IX, a HO-1 inhibitor. Notably, our findings provide the first evidence that HO-1 inhibition in combination with PLX4032 under physiological oxygen tension and hypoxia restores and increases the expression of the NK ligands ULBP3 and B7H6 compared to cells exposed to PLX4032 alone. Interestingly, although silencing NRF2 prevented PLX4032 induction of HO-1, other NRF2 targeted genes were unaffected, highlighting a pivotal role of HO-1 in melanoma resistance and immune escape. The present findings may enhance translation and highlight the potential of the HO-1 inhibitors in the therapy of BRAFV600 melanomas.
Keywords: HO-1; NK ligands; NRF2; hypoxia; melanoma; oxygen tension; physiological normoxia; response and/or resistance to therapy; target therapy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
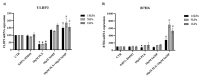
References
-
- Marengo B., Nitti M., Furfaro A.L., Colla R., Ciucis C.D., Marinari U.M., Pronzato M.A., Traverso N., Domenicotti C. Redox Homeostasis and Cellular Antioxidant Systems: Crucial Players in Cancer Growth and Therapy. Oxid. Med. Cell. Longev. 2016;2016:6235641. doi: 10.1155/2016/6235641. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

