Immunomodulatory Properties of Human Breast Milk: MicroRNA Contents and Potential Epigenetic Effects
- PMID: 35740242
- PMCID: PMC9219990
- DOI: 10.3390/biomedicines10061219
Immunomodulatory Properties of Human Breast Milk: MicroRNA Contents and Potential Epigenetic Effects
Abstract
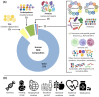
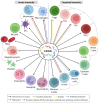
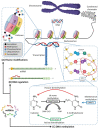
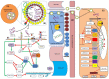
Infants who are exclusively breastfed in the first six months of age receive adequate nutrients, achieving optimal immune protection and growth. In addition to the known nutritional components of human breast milk (HBM), i.e., water, carbohydrates, fats and proteins, it is also a rich source of microRNAs, which impact epigenetic mechanisms. This comprehensive work presents an up-to-date overview of the immunomodulatory constituents of HBM, highlighting its content of circulating microRNAs. The epigenetic effects of HBM are discussed, especially those regulated by miRNAs. HBM contains more than 1400 microRNAs. The majority of these microRNAs originate from the lactating gland and are based on the remodeling of cells in the gland during breastfeeding. These miRNAs can affect epigenetic patterns by several mechanisms, including DNA methylation, histone modifications and RNA regulation, which could ultimately result in alterations in gene expressions. Therefore, the unique microRNA profile of HBM, including exosomal microRNAs, is implicated in the regulation of the genes responsible for a variety of immunological and physiological functions, such as FTO, INS, IGF1, NRF2, GLUT1 and FOXP3 genes. Hence, studying the HBM miRNA composition is important for improving the nutritional approaches for pregnancy and infant's early life and preventing diseases that could occur in the future. Interestingly, the composition of miRNAs in HBM is affected by multiple factors, including diet, environmental and genetic factors.
Keywords: DNA methylation; RNA regulation; breastfeeding; epigenetics; histone modification; lactation; miRNA.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Infant and Young Child Nutrition: Global Strategy on Infant and Young Child Feeding. [(accessed on 10 March 2022)]. Available online: https://apps.who.int/gb/archive/pdf_files/WHA55/ea5515.pdf.
-
- Boronat-Catalá M., Montiel-Company J.M., Bellot-Arcís C., Almerich-Silla J.M., Catalá-Pizarro M. Association between duration of breastfeeding and malocclusions in primary and mixed dentition: A systematic review and meta-analysis. Sci. Rep. 2017;7:5048. doi: 10.1038/s41598-017-05393-y. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

