Thrombotic Complications after COVID-19 Vaccination: Diagnosis and Treatment Options
- PMID: 35740269
- PMCID: PMC9220036
- DOI: 10.3390/biomedicines10061246
Thrombotic Complications after COVID-19 Vaccination: Diagnosis and Treatment Options
Abstract
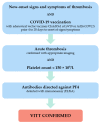
Coronavirus disease 2019 (COVID-19) vaccines were developed a few months after the emergence of the pandemic. The first cases of vaccine-induced thrombotic complications after the use of adenoviral vector vaccines ChAdOx1 nCoV-19 by AstraZeneca, and Ad26.COV2.S by Johnson & Johnson/Janssen, were announced shortly after the initiation of a global vaccination program. In these cases, the occurrence of thrombotic events at unusual sites-predominantly located in the venous vascular system-in association with concomitant thrombocytopenia were observed. Since this new entity termed vaccine-induced thrombotic thrombocytopenia (VITT) shows similar pathophysiologic mechanisms as heparin-induced thrombocytopenia (HIT), including the presence of antibodies against heparin/platelet factor 4 (PF4), standard routine treatment for thrombotic events-arterial or venous-are not appropriate and may also cause severe harm in affected patients. Thrombotic complications were also rarely documented after vaccination with mRNA vaccines, but a typical VITT phenomenon has, to date, not been established for these vaccines. The aim of this review is to give a concise and feasible overview of diagnostic and therapeutic strategies in COVID-19 vaccine-induced thrombotic complications.
Keywords: (autoimmune) heparin-induced thrombocytopenia (HIT); adenoviral vector vaccines; anticoagulation; coronavirus disease 2019 (COVID-19); severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2); thrombosis; thrombosis thrombocytopenia syndrome (TTS); vaccination; vaccine-induced thrombotic thrombocytopenia (VITT).
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Schultz N.H., Sorvoll I.H., Michelsen A.E., Munthe L.A., Lund-Johansen F., Ahlen M.T., Wiedmann M., Aamodt A.H., Skattor T.H., Tjonnfjord G.E., et al. Thrombosis and Thrombocytopenia after ChAdOx1 nCoV-19 Vaccination. N. Engl. J. Med. 2021;384:2124–2130. doi: 10.1056/NEJMoa2104882. - DOI - PMC - PubMed
-
- See I., Su J.R., Lale A., Woo E.J., Guh A.Y., Shimabukuro T.T., Streiff M.B., Rao A.K., Wheeler A.P., Beavers S.F., et al. US Case Reports of Cerebral Venous Sinus Thrombosis With Thrombocytopenia After Ad26.COV2.S Vaccination, March 2 to April 21, 2021. JAMA. 2021;325:2448–2456. doi: 10.1001/jama.2021.7517. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous