Comprehensive Analysis of Disease Pathology in Immunocompetent and Immunocompromised Hosts following Pulmonary SARS-CoV-2 Infection
- PMID: 35740365
- PMCID: PMC9219777
- DOI: 10.3390/biomedicines10061343
Comprehensive Analysis of Disease Pathology in Immunocompetent and Immunocompromised Hosts following Pulmonary SARS-CoV-2 Infection
Abstract
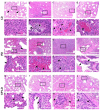
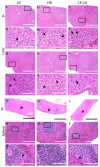
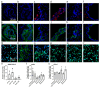
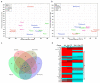
The Coronavirus disease 2019 (COVID-19) pandemic disproportionately affects immunocompetent and immunocompromised individuals, with the latter group being more vulnerable to severe disease and death. However, the differential pathogenesis of SARS-CoV-2 in the context of a specific immunological niche remains unknown. Similarly, systematic analysis of disease pathology in various extrapulmonary organs in immunocompetent and immunocompromised hosts during SARS-CoV-2 infection is not fully understood. We used a hamster model of SARS-CoV-2 infection, which recapitulates the pathophysiology of patients with mild-to-moderate COVID-19, to determine the dynamics of SARS-CoV-2 replication and histopathology at organ-level niches and map how COVID-19 symptoms vary in different immune contexts. Hamsters were intranasally infected with low (LD) or high (HD) inoculums of SARS-CoV-2, and the kinetics of disease pathology and viral load in multiple organs, antibody response, inflammatory cytokine expression, and genome-wide lung transcriptome by RNAseq analysis were determined and compared against corresponding responses from chemically induced immunocompromised hamsters. We observed transient body weight loss proportional to the SARS-CoV-2 infectious dose in immunocompetent hamsters. The kinetics of viral replication and peak viral loads were similar between LD and HD groups, although the latter developed more severe disease pathology in organs. Both groups generated a robust serum antibody response. In contrast, infected immunocompromised animals showed more prolonged body weight loss and mounted an inadequate SARS-CoV-2-neutralizing antibody response. The live virus was detected in the pulmonary and extrapulmonary organs for extended periods. These hamsters also had persistent inflammation with severe bronchiolar-alveolar hyperplasia/metaplasia. Consistent with the differential disease presentation, distinct changes in inflammation and immune cell response pathways and network gene expression were seen in the lungs of SARS-CoV-2-infected immunocompetent and immunocompromised animals.
Keywords: COVID-19; RNAseq; animal models; antibody; extrapulmonary; gene networks; immune suppression; immunopathology; thrombosis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






Similar articles
-
Coinfection by Severe Acute Respiratory Syndrome Coronavirus 2 and Influenza A(H1N1)pdm09 Virus Enhances the Severity of Pneumonia in Golden Syrian Hamsters.Clin Infect Dis. 2021 Jun 15;72(12):e978-e992. doi: 10.1093/cid/ciaa1747. Clin Infect Dis. 2021. PMID: 33216851 Free PMC article.
-
Simulation of the Clinical and Pathological Manifestations of Coronavirus Disease 2019 (COVID-19) in a Golden Syrian Hamster Model: Implications for Disease Pathogenesis and Transmissibility.Clin Infect Dis. 2020 Dec 3;71(9):2428-2446. doi: 10.1093/cid/ciaa325. Clin Infect Dis. 2020. PMID: 32215622 Free PMC article.
-
Characterization of Virus Replication, Pathogenesis, and Cytokine Responses in Syrian Hamsters Inoculated with SARS-CoV-2.J Inflamm Res. 2021 Aug 11;14:3781-3795. doi: 10.2147/JIR.S323026. eCollection 2021. J Inflamm Res. 2021. PMID: 34408462 Free PMC article.
-
Pathogenesis-directed therapy of 2019 novel coronavirus disease.J Med Virol. 2021 Mar;93(3):1320-1342. doi: 10.1002/jmv.26610. Epub 2020 Nov 10. J Med Virol. 2021. PMID: 33073355 Review.
-
Therapeutic implications of ongoing alveolar viral replication in COVID-19.Lancet Rheumatol. 2022 Feb;4(2):e135-e144. doi: 10.1016/S2665-9913(21)00322-2. Epub 2021 Dec 1. Lancet Rheumatol. 2022. PMID: 34873587 Free PMC article. Review.
Cited by
-
Differences in incidence, nature of symptoms, and duration of long COVID among hospitalised migrant and non-migrant patients in the Netherlands: a retrospective cohort study.Lancet Reg Health Eur. 2023 Apr 7;29:100630. doi: 10.1016/j.lanepe.2023.100630. eCollection 2023 Jun. Lancet Reg Health Eur. 2023. PMID: 37261215 Free PMC article.
-
The effect of molnupiravir and nirmatrelvir on SARS-CoV-2 genome diversity in severe models of COVID-19.bioRxiv [Preprint]. 2024 Dec 20:2024.02.27.582110. doi: 10.1101/2024.02.27.582110. bioRxiv. 2024. Update in: Microbiol Spectr. 2025 May 6;13(5):e0182924. doi: 10.1128/spectrum.01829-24. PMID: 38464327 Free PMC article. Updated. Preprint.
-
Characteristics of animal models for COVID-19.Animal Model Exp Med. 2022 Oct;5(5):401-409. doi: 10.1002/ame2.12278. Animal Model Exp Med. 2022. PMID: 36301011 Free PMC article. Review.
-
The effect of molnupiravir and nirmatrelvir on SARS-CoV-2 genome diversity in severe models of COVID-19.Microbiol Spectr. 2025 May 6;13(5):e0182924. doi: 10.1128/spectrum.01829-24. Epub 2025 Mar 25. Microbiol Spectr. 2025. PMID: 40130852 Free PMC article.
-
Implementing Translational Research to Understand the Future of COVID-19 and Its Long-Term Consequences: A Degrowth Perspective or the Transformation of a Global Emergency?Biomedicines. 2023 Jan 3;11(1):117. doi: 10.3390/biomedicines11010117. Biomedicines. 2023. PMID: 36672625 Free PMC article.
References
-
- WHO Coronavirus (COVID-19) Dashboard. [(accessed on 12 April 2022)]. Available online: https://covid19.who.int/
-
- Hadjadj J., Yatim N., Barnabei L., Corneau A., Boussier J., Smith N., Pere H., Charbit B., Bondet V., Chenevier-Gobeaux C., et al. Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients. Science. 2020;369:718–724. doi: 10.1126/science.abc6027. - DOI - PMC - PubMed
-
- Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z., Xiang J., Wang Y., Song B., Gu X., et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

