Overview and Update on Extracellular Vesicles: Considerations on Exosomes and Their Application in Modern Medicine
- PMID: 35741325
- PMCID: PMC9220244
- DOI: 10.3390/biology11060804
Overview and Update on Extracellular Vesicles: Considerations on Exosomes and Their Application in Modern Medicine
Abstract
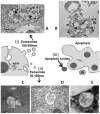
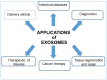
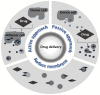
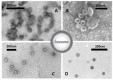
In recent years, there has been a rapid growth in the knowledge of cell-secreted extracellular vesicle functions. They are membrane enclosed and loaded with proteins, nucleic acids, lipids, and other biomolecules. After being released into the extracellular environment, some of these vesicles are delivered to recipient cells; consequently, the target cell may undergo physiological or pathological changes. Thus, extracellular vesicles as biological nano-carriers, have a pivotal role in facilitating long-distance intercellular communication. Understanding the mechanisms that mediate this communication process is important not only for basic science but also in medicine. Indeed, extracellular vesicles are currently seen with immense interest in nanomedicine and precision medicine for their potential use in diagnostic, prognostic, and therapeutic applications. This paper aims to summarize the latest advances in the study of the smallest subtype among extracellular vesicles, the exosomes. The article is divided into several sections, focusing on exosomes' nature, characteristics, and commonly used strategies and methodologies for their separation, characterization, and visualization. By searching an extended portion of the relevant literature, this work aims to give a quick outline of advances in exosomes' extensive nanomedical applications. Moreover, considerations that require further investigations before translating them to clinical applications are summarized.
Keywords: drug delivery systems; exosomes characterization; extracellular vesicles; nanomedicine.
Conflict of interest statement
The author declares no conflict of interest.
Figures





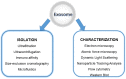
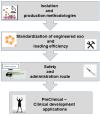
References
-
- Drexler E.K., Peterson C., Pergamit G. Unbounding the Future: The Nanothechnology Revolution. William Morrow and Company, Inc.; New York, NY, USA: 1991.
-
- Chidambaran M., Manavalan R., Kathiresan K. Nanotherapeutics to overcome conventional cancer chemotherapy limitations. J. Pharm. Sci. 2011;14:67–77. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

