Targeting the Cation-Chloride Co-Transporter NKCC1 to Re-Establish GABAergic Inhibition and an Appropriate Excitatory/Inhibitory Balance in Selective Neuronal Circuits: A Novel Approach for the Treatment of Alzheimer's Disease
- PMID: 35741668
- PMCID: PMC9221351
- DOI: 10.3390/brainsci12060783
Targeting the Cation-Chloride Co-Transporter NKCC1 to Re-Establish GABAergic Inhibition and an Appropriate Excitatory/Inhibitory Balance in Selective Neuronal Circuits: A Novel Approach for the Treatment of Alzheimer's Disease
Abstract
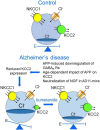
GABA, the main inhibitory neurotransmitter in the adult brain, depolarizes and excites immature neurons because of an initially higher intracellular chloride concentration [Cl-]i due to the delayed expression of the chloride exporter KCC2 at birth. Depolarization-induced calcium rise via NMDA receptors and voltage-dependent calcium channels is instrumental in shaping neuronal circuits and in controlling the excitatory (E)/inhibitory (I) balance in selective brain areas. An E/I imbalance accounts for cognitive impairment observed in several neuropsychiatric disorders. The aim of this review is to summarize recent data on the mechanisms by which alterations of GABAergic signaling alter the E/I balance in cortical and hippocampal neurons in Alzheimer's disease (AD) and the role of cation-chloride co-transporters in this process. In particular, we discuss the NGF and AD relationship and how mice engineered to express recombinant neutralizing anti-NGF antibodies (AD11 mice), which develop a neurodegenerative pathology reminiscent of that observed in AD patients, exhibit a depolarizing action of GABA due to KCC2 impairment. Treating AD and other forms of dementia with bumetanide, a selective KCC2 antagonist, contributes to re-establishing a proper E/I balance in selective brain areas, leading to amelioration of AD symptoms and the slowing down of disease progression.
Keywords: AD11 transgenic mice; Alzheimer’s disease; KCC2 dysfunction; NGF; bumetanide treatment; cation-chloride co-transporters; depolarizing GABAA-mediated neurotransmission.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
Publication types
LinkOut - more resources
Full Text Sources

