Differentially Expressed miRNAs in Age-Related Neurodegenerative Diseases: A Meta-Analysis
- PMID: 35741796
- PMCID: PMC9222420
- DOI: 10.3390/genes13061034
Differentially Expressed miRNAs in Age-Related Neurodegenerative Diseases: A Meta-Analysis
Abstract
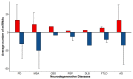
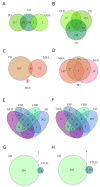
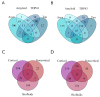
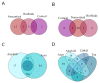
To date, no neurodegenerative diseases (NDDs) have cures, and the underlying mechanism of their pathogenesis is undetermined. As miRNAs extensively regulate all biological processes and are crucial regulators of healthy brain function, miRNAs differentially expressed in NDDs may provide insight into the factors that contribute to the emergence of protein inclusions and the propagation of deleterious cellular environments. A meta-analysis of miRNAs dysregulated in Alzheimer's disease, Parkinson's disease, multiple system atrophy, progressive supranuclear palsy, corticobasal degeneration, dementia with Lewy bodies and frontotemporal lobar degeneration (TDP43 variant) was performed to determine if diseases within a proteinopathy have distinct or shared mechanisms of action leading to neuronal death, and if proteinopathies can be classified on the basis of their miRNA profiles. Our results identified both miRNAs distinct to the anatomy, disease type and pathology, and miRNAs consistently dysregulated within single proteinopathies and across neurodegeneration in general. Our results also highlight the necessity to minimize the variability between studies. These findings showcase the need for more transcriptomic research on infrequently occurring NDDs, and the need for the standardization of research techniques and platforms utilized across labs and diseases.
Keywords: amyloidopathies; miRNAs; neurodegenerative diseases; synucleinopathies; tauopathies.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Motor neuron TDP-43 proteinopathy in progressive supranuclear palsy and corticobasal degeneration.Brain. 2022 Aug 27;145(8):2769-2784. doi: 10.1093/brain/awac091. Brain. 2022. PMID: 35274674
-
Phosphorylated Smad 2/3 colocalizes with phospho-tau inclusions in Pick disease, progressive supranuclear palsy, and corticobasal degeneration but not with alpha-synuclein inclusions in multiple system atrophy or dementia with Lewy bodies.J Neuropathol Exp Neurol. 2007 Nov;66(11):1019-26. doi: 10.1097/nen.0b013e31815885ad. J Neuropathol Exp Neurol. 2007. PMID: 17984683
-
Filamentous nerve cell inclusions in neurodegenerative diseases: tauopathies and alpha-synucleinopathies.Philos Trans R Soc Lond B Biol Sci. 1999 Jun 29;354(1386):1101-18. doi: 10.1098/rstb.1999.0466. Philos Trans R Soc Lond B Biol Sci. 1999. PMID: 10434313 Free PMC article. Review.
-
Incidence and morphology of secondary TDP-43 proteinopathies: Part 1.Folia Neuropathol. 2022;60(3):267-276. doi: 10.5114/fn.2022.120314. Folia Neuropathol. 2022. PMID: 36382478 Review.
-
Diffusion tensor magnetic resonance imaging for single subject diagnosis in neurodegenerative diseases.Brain. 2013 Jul;136(Pt 7):2253-61. doi: 10.1093/brain/awt118. Epub 2013 May 31. Brain. 2013. PMID: 23729473
Cited by
-
Cerebellar Micro-RNA Profile in a Mouse Model of Spinocerebellar Ataxia Type 2.Neurol Genet. 2024 Mar 28;10(2):e200144. doi: 10.1212/NXG.0000000000200144. eCollection 2024 Apr. Neurol Genet. 2024. PMID: 38715656 Free PMC article.
-
Differential Expression of MicroRNAs and Predicted Drug Target in Amyotrophic Lateral Sclerosis.J Mol Neurosci. 2023 Jun;73(6):375-390. doi: 10.1007/s12031-023-02124-z. Epub 2023 May 30. J Mol Neurosci. 2023. PMID: 37249795
-
Epigenetic Modifications Associated With Wildland-Urban Interface (WUI) Firefighting.Environ Mol Mutagen. 2025 Jan-Feb;66(1-2):22-33. doi: 10.1002/em.70002. Epub 2025 Feb 19. Environ Mol Mutagen. 2025. PMID: 39968828 Free PMC article.
-
Staufen2 dysregulation in neurodegenerative disease.J Biol Chem. 2025 Mar;301(3):108316. doi: 10.1016/j.jbc.2025.108316. Epub 2025 Feb 13. J Biol Chem. 2025. PMID: 39955058 Free PMC article.
-
Perspective Strategies for Interventions in Parkinsonism: Remedying the Neglected Role of TPPP.Cells. 2024 Feb 14;13(4):338. doi: 10.3390/cells13040338. Cells. 2024. PMID: 38391951 Free PMC article.
References
-
- Collaborators G.U.N.D., Feigin V.L., Vos T., Alahdab F., Amit A.M.L., Barnighausen T.W., Beghi E., Beheshti M., Chavan P.P., Criqui M.H., et al. Burden of Neurological Disorders across the US From 1990–2017: A global burden of disease study. JAMA Neurol. 2021;78:165–176. doi: 10.1001/jamaneurol.2020.4152. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

