The Development of FAK Inhibitors: A Five-Year Update
- PMID: 35742823
- PMCID: PMC9223874
- DOI: 10.3390/ijms23126381
The Development of FAK Inhibitors: A Five-Year Update
Abstract
Focal adhesion kinase (FAK) is a non-receptor tyrosine kinase over-expressed in different solid cancers. In recent years, FAK has been recognized as a new target for the development of antitumor agents, useful to contrast tumor development and metastasis formation. To date, studies on the role of FAK and FAK inhibitors are of great interest for both pharmaceutical companies and academia. This review is focused on compounds able to block FAK with different potencies and with different mechanisms of action, that have appeared in the literature since 2017. Furthermore, new emerging PROTAC molecules have appeared in the literature. This summary could improve knowledge of new FAK inhibitors and provide information for future investigations, in particular, from a medicinal chemistry point of view.
Keywords: FAK inhibitors; PROTAC; anticancer compounds; focal adhesion kinase; medicinal chemistry; pyrimidines; triazines.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
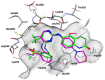
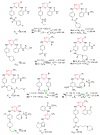
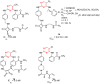
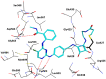
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

