Neuroprotective Effect of Bcl-2 on Lipopolysaccharide-Induced Neuroinflammation in Cortical Neural Stem Cells
- PMID: 35742844
- PMCID: PMC9223771
- DOI: 10.3390/ijms23126399
Neuroprotective Effect of Bcl-2 on Lipopolysaccharide-Induced Neuroinflammation in Cortical Neural Stem Cells
Abstract
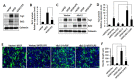
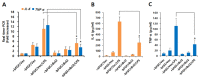
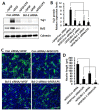
Neuroinflammation is involved in the pathogenesis of neurodegenerative diseases due to increased levels of pro-inflammatory cytokines in the central nervous system (CNS). Chronic neuroinflammation induced by neurotoxic molecules accelerates neuronal damage. B-cell lymphoma 2 (Bcl-2) is generally accepted to be an important anti-apoptotic factor. However, the role of Bcl-2 in neuroprotection against neuroinflammation remains to be determined. The purpose of this study was to investigate the neuroprotective effect of Bcl-2 on lipopolysaccharide (LPS)-induced neuroinflammation in cortical neural stem cells (NSCs). LPS decreased mRNA and protein levels of Tuj-1, a neuron marker, and also suppressed neurite outgrowth, indicating that LPS results in inhibition of neuronal differentiation of NSCs. Furthermore, LPS treatment inhibited Bcl-2 expression during neuronal differentiation; inhibition of neuronal differentiation by LPS was rescued by Bcl-2 overexpression. LPS-induced pro-inflammatory cytokines, including interleukin (IL)-6 and tumor necrosis factor alpha (TNF-α), were decreased by Bcl-2 overexpression. Conversely, Bcl-2 siRNA increased the LPS-induced levels of IL-6 and TNF-α, and decreased neuronal differentiation of NSCs, raising the possibility that Bcl-2 mediates neuronal differentiation by inhibiting the LPS-induced inflammatory response in NSC. These results suggest that Bcl-2 has a neuroprotective effect by inhibiting the LPS-induced inflammatory response in NSCs.
Keywords: B-cell lymphoma 2; lipopolysaccharide; neural stem cells; neuroinflammation; neuronal differentiation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

