Coronin1C Is a GDP-Specific Rab44 Effector That Controls Osteoclast Formation by Regulating Cell Motility in Macrophages
- PMID: 35743062
- PMCID: PMC9224296
- DOI: 10.3390/ijms23126619
Coronin1C Is a GDP-Specific Rab44 Effector That Controls Osteoclast Formation by Regulating Cell Motility in Macrophages
Abstract
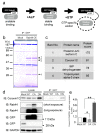
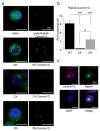
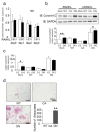
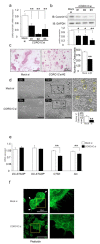
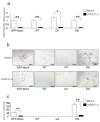
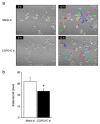
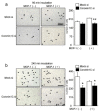
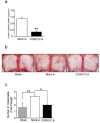
Osteoclasts are multinucleated bone-resorbing cells that are formed by the fusion of macrophages. Recently, we identified Rab44, a large Rab GTPase, as an upregulated gene during osteoclast differentiation that negatively regulates osteoclast differentiation. However, the molecular mechanisms by which Rab44 negatively regulates osteoclast differentiation remain unknown. Here, we found that the GDP form of Rab44 interacted with the actin-binding protein, Coronin1C, in murine macrophages. Immunoprecipitation experiments revealed that the interaction of Rab44 and Coronin1C occurred in wild-type and a dominant-negative (DN) mutant of Rab44, but not in a constitutively active (CA) mutant of Rab44. Consistent with these findings, the expression of the CA mutant inhibited osteoclast differentiation, whereas that of the DN mutant enhanced this differentiation. Using a phase-contrast microscope, Coronin1C-knockdown osteoclasts apparently impaired multinuclear formation. Moreover, Coronin1C knockdown impaired the migration and chemotaxis of RAW-D macrophages. An in vivo experimental system demonstrated that Coronin1C knockdown suppresses osteoclastogenesis. Therefore, the decreased cell formation and fusion of Coronin1C-depleted osteoclasts might be due to the decreased migration of Coronin1C-knockdown macrophages. These results indicate that Coronin1C is a GDP-specific Rab44 effector that controls osteoclast formation by regulating cell motility in macrophages.
Keywords: Coronin1C; Rab44; cell motility; effector; osteoclast differentiation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Takayanagi H., Kim S., Koga T., Nishina H., Isshiki M., Yoshida H., Saiura A., Isobe M., Yokochi T., Inoue J., et al. Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev. Cell. 2002;3:889–901. doi: 10.1016/S1534-5807(02)00369-6. - DOI - PubMed
-
- Okusha Y., Tran M.T., Itagaki M., Sogawa C., Eguchi T., Okui T., Kadowaki T., Sakai E., Tsukuba T., Okamoto K. Rab11A Functions as a Negative Regulator of Osteoclastogenesis through Dictating Lysosome-Induced Proteolysis of c-fms and RANK Surface Receptors. Cells. 2020;9:2384. doi: 10.3390/cells9112384. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

