Kinase Activity of PAR1b, Which Mediates Nuclear Translocation of the BRCA1 Tumor Suppressor, Is Potentiated by Nucleic Acid-Mediated PAR1b Multimerization
- PMID: 35743080
- PMCID: PMC9223676
- DOI: 10.3390/ijms23126634
Kinase Activity of PAR1b, Which Mediates Nuclear Translocation of the BRCA1 Tumor Suppressor, Is Potentiated by Nucleic Acid-Mediated PAR1b Multimerization
Abstract
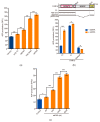
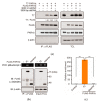
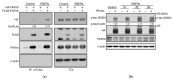
PAR1b is a cytoplasmic serine/threonine kinase that controls cell polarity and cell-cell interaction by regulating microtubule stability while mediating cytoplasmic-to-nuclear translocation of BRCA1. PAR1b is also a cellular target of the CagA protein of Helicobacter pylori, which leads to chronic infection causatively associated with the development of gastric cancer. The CagA-PAR1b interaction inactivates the kinase activity of PAR1b and thereby dampens PAR1b-mediated BRCA1 phosphorylation, which reduces the level of nuclear BRCA1 and thereby leads to BRCAness and BRCAness-associated genome instability underlying gastric carcinogenesis. While PAR1b can multimerize within the cells, little is known about the mechanism and functional role of PAR1b multimerization. We found in the present study that PAR1b was multimerized in vitro by binding with nucleic acids (both single- and double-stranded DNA/RNA) via the spacer region in a manner independent of nucleic-acid sequences, which markedly potentiated the kinase activity of PAR1b. Consistent with these in vitro observations, cytoplasmic introduction of double-stranded DNA or expression of single-stranded RNA increased the PAR1b kinase activity in the cells. These findings indicate that the cytoplasmic DNA/RNA contribute to nuclear accumulation of BRCA1 by constitutively activating/potentiating cytoplasmic PAR1b kinase activity, which is subverted in gastric epithelial cells upon delivery of H. pylori CagA oncoprotein.
Keywords: EBER; PAR1b; gastric cancer; kinase activation; multimerization; nucleic acids.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Helicobacter pylori CagA elicits BRCAness to induce genome instability that may underlie bacterial gastric carcinogenesis.Cell Host Microbe. 2021 Jun 9;29(6):941-958.e10. doi: 10.1016/j.chom.2021.04.006. Epub 2021 May 13. Cell Host Microbe. 2021. PMID: 33989515
-
Impact of structural polymorphism for the Helicobacter pylori CagA oncoprotein on binding to polarity-regulating kinase PAR1b.Sci Rep. 2016 Jul 22;6:30031. doi: 10.1038/srep30031. Sci Rep. 2016. PMID: 27445265 Free PMC article.
-
Structural and functional diversity in the PAR1b/MARK2-binding region of Helicobacter pylori CagA.Cancer Sci. 2008 Oct;99(10):2004-11. doi: 10.1111/j.1349-7006.2008.00950.x. Cancer Sci. 2008. PMID: 19016760 Free PMC article.
-
Helicobacter pylori-induced DNA double-stranded break in the development of gastric cancer.Cancer Sci. 2022 Jun;113(6):1909-1918. doi: 10.1111/cas.15357. Epub 2022 Apr 20. Cancer Sci. 2022. PMID: 35359025 Free PMC article. Review.
-
Helicobacter pylori and gastric carcinogenesis.J Gastroenterol. 2009;44(4):239-48. doi: 10.1007/s00535-009-0014-1. Epub 2009 Mar 7. J Gastroenterol. 2009. PMID: 19271114 Review.
Cited by
-
Role of Exosomes and Their Potential as Biomarkers in Epstein-Barr Virus-Associated Gastric Cancer.Cancers (Basel). 2023 Jan 12;15(2):469. doi: 10.3390/cancers15020469. Cancers (Basel). 2023. PMID: 36672418 Free PMC article. Review.
References
-
- Imai S., Ooki T., Murata-Kamiya N., Komura D., Tahmina K., Wu W., Takahashi-Kanemitsu A., Knight C.T., Kunita A., Suzuki N., et al. Helicobacter pylori CagA Elicits BRCAness to Induce Genome Instability That May Underlie Bacterial Gastric Carcinogenesis. Cell Host Microbe. 2021;29:941–958. doi: 10.1016/j.chom.2021.04.006. - DOI - PubMed
-
- Suzuki A., Hirata M., Kamimura K., Maniwa R., Yamanaka T., Mizuno K., Kishikawa M., Hirose H., Amano Y., Izumi N., et al. aPKC Acts Upstream of PAR-1b in Both the Establishment and Maintenance of Mammalian Epithelial Polarity. Curr. Biol. 2004;14:1425–1435. doi: 10.1016/j.cub.2004.08.021. - DOI - PubMed
-
- Drewes G., Trinczek B., Illenberger S., Biernat J., Schmitt-Ulms G., Meyer H.E., Mandelkow E.-M., Mandelkow E. Microtubule-Associated Protein/Microtubule Affinity-Regulating Kinase (P110mark): A Novel Protein Kinase That Regulates Tau-Microtubule Interactions and Dynamic Instability by Phosphorylation at the Alzheimer-Specific Site Serine 262. J. Biol. Chem. 1995;270:7679–7688. doi: 10.1074/jbc.270.13.7679. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

