Inspired by Nature-Functional Analogues of Molybdenum and Tungsten-Dependent Oxidoreductases
- PMID: 35744820
- PMCID: PMC9227248
- DOI: 10.3390/molecules27123695
Inspired by Nature-Functional Analogues of Molybdenum and Tungsten-Dependent Oxidoreductases
Abstract
Throughout the previous ten years many scientists took inspiration from natural molybdenum and tungsten-dependent oxidoreductases to build functional active site analogues. These studies not only led to an ever more detailed mechanistic understanding of the biological template, but also paved the way to atypical selectivity and activity, such as catalytic hydrogen evolution. This review is aimed at representing the last decade's progress in the research of and with molybdenum and tungsten functional model compounds. The portrayed systems, organized according to their ability to facilitate typical and artificial enzyme reactions, comprise complexes with non-innocent dithiolene ligands, resembling molybdopterin, as well as entirely non-natural nitrogen, oxygen, and/or sulfur bearing chelating donor ligands. All model compounds receive individual attention, highlighting the specific novelty that each provides for our understanding of the enzymatic mechanisms, such as oxygen atom transfer and proton-coupled electron transfer, or that each presents for exploiting new and useful catalytic capability. Overall, a shift in the application of these model compounds towards uncommon reactions is noted, the latter are comprehensively discussed.
Keywords: OAT; acetylene hydratase; dithiolene; functional models; hydrogen evolution; oxidoreductases.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
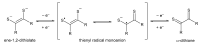


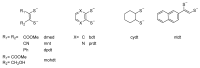
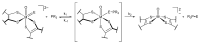
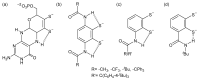




References
-
- Majumdar A., Sarkar S. Bioinorganic chemistry of molybdenum and tungsten enzymes: A structural-functional modeling approach. Coord. Chem. Rev. 2011;255:1039–1054. doi: 10.1016/j.ccr.2010.11.027. - DOI
-
- Hine F.J., Taylor A.J., Garner C.D. Dithiolene complexes and the nature of molybdopterin. Coord. Chem. Rev. 2010;254:1570–1579. doi: 10.1016/j.ccr.2010.01.017. - DOI
-
- Stiefel E.I., editor. Dithiolene Chemistry: Synthesis, Properties, and Applications. John Wiley & Sons, Inc.; Hoboken, NJ, USA: 2003.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

