Bacterial Ghosts of Pseudomonas aeruginosa as a Promising Candidate Vaccine and Its Application in Diabetic Rats
- PMID: 35746518
- PMCID: PMC9228170
- DOI: 10.3390/vaccines10060910
Bacterial Ghosts of Pseudomonas aeruginosa as a Promising Candidate Vaccine and Its Application in Diabetic Rats
Abstract
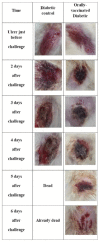
Infections with Pseudomonas aeruginosa (PA) pose a major clinical threat worldwide especially to immunocompromised patients. As a novel vaccine network for many kinds of bacteria, bacterial ghosts (BGs) have recently been introduced. In the present research, using Sponge-Like Reduced Protocol, P. aeruginosa ghosts (PAGs) were prepared to maintain surface antigens and immunogenicity. This is the first study, to our knowledge, on the production of chemically induced well-structured bacterial ghosts for PA using concentrations of different chemicals. The research was carried out using diabetic rats who were orally immunized at two-week intervals with three doses of PAGs. Rats were subsequently challenged either by the oral route or by the model of ulcer infection with PA. In challenged rats, in addition to other immunological parameters, organ bioburden and wound healing were determined, respectively. Examination of the scanning and transmission electron microscope (EM) proved that PAGs with a proper three-dimensional structure were obtained. In contrast to control groups, oral PAGs promoted the generation of agglutinating antibodies, the development of IFN-γ, and the increase in phagocytic activity in vaccinated groups. Antibodies of the elicited PAGs were reactive to PA proteins and lipopolysaccharides. The defense against the PA challenge was observed in PAGs-immunized diabetic rats. The resulting PAGs in orally vaccinated diabetic rats were able to evoke unique humoral and cell-mediated immune responses and to defend them from the threat of skin wound infection. These results have positive implications for future studies on the PA vaccine.
Keywords: Pseudomonas aeruginosa; bacterial ghost; diabetic ulcer; oral immunization; sponge-like reduced protocol (SLRP); vaccine.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






Similar articles
-
Generation of a novel Streptococcus agalactiae ghost vaccine and examination of its immunogenicity against virulent challenge in tilapia.Fish Shellfish Immunol. 2018 Oct;81:49-56. doi: 10.1016/j.fsi.2018.06.055. Epub 2018 Jun 30. Fish Shellfish Immunol. 2018. PMID: 29969706
-
Preparation of porcine enterotoxigenic Escherichia coli (ETEC) ghosts and immunogenic analysis in a mouse model.Microb Pathog. 2019 Jan;126:224-230. doi: 10.1016/j.micpath.2018.11.015. Epub 2018 Nov 11. Microb Pathog. 2019. PMID: 30428380
-
Incorporation of membrane-anchored flagellin into Salmonella Gallinarum bacterial ghosts induces early immune responses and protection against fowl typhoid in young layer chickens.Vet Immunol Immunopathol. 2018 May;199:61-69. doi: 10.1016/j.vetimm.2018.03.011. Epub 2018 Mar 29. Vet Immunol Immunopathol. 2018. PMID: 29678231
-
New strategies for combination vaccines based on the extended recombinant bacterial ghost system.Vaccine. 1999 Mar 26;17(13-14):1643-9. doi: 10.1016/s0264-410x(98)00423-x. Vaccine. 1999. PMID: 10194817 Review.
-
Pseudomonas aeruginosa antigens as potential vaccines.FEMS Microbiol Rev. 1997 Nov;21(3):243-77. doi: 10.1111/j.1574-6976.1997.tb00353.x. FEMS Microbiol Rev. 1997. PMID: 9451816 Review.
Cited by
-
Pseudomonas aeruginosa Vaccine Development: Lessons, Challenges, and Future Innovations.Int J Mol Sci. 2025 Feb 25;26(5):2012. doi: 10.3390/ijms26052012. Int J Mol Sci. 2025. PMID: 40076637 Free PMC article. Review.
-
Proteomic approach to identify host cell attachment proteins provides protective Pseudomonas aeruginosa vaccine antigen FtsZ.NPJ Vaccines. 2024 Oct 28;9(1):204. doi: 10.1038/s41541-024-00994-x. NPJ Vaccines. 2024. PMID: 39468053 Free PMC article.
-
Pseudomonas aeruginosa: Recent Advances in Vaccine Development.Vaccines (Basel). 2022 Jul 8;10(7):1100. doi: 10.3390/vaccines10071100. Vaccines (Basel). 2022. PMID: 35891262 Free PMC article. Review.
-
Bacteriophage-encoded protein utilization in bacterial ghost production: a mini-review.World J Microbiol Biotechnol. 2024 Jul 29;40(9):284. doi: 10.1007/s11274-024-04091-y. World J Microbiol Biotechnol. 2024. PMID: 39073496 Review.
References
-
- Ruiz-Garbajosa P., Cantón R. Epidemiology of antibiotic resistance in Pseudomonas aeruginosa. Implications for empiric and definitive therapy. Rev. Esp. Quimioter. 2017;1:8–12. - PubMed
-
- Wahab W.F.A. Diabetic Foot Infections with Pseudomonas: Jabir Abueliz Diabetic Center Khartoum Experience. Clin. Res. Foot Ankle. 2014;s3 doi: 10.4172/2329-910x.s3-001. - DOI
LinkOut - more resources
Full Text Sources
Research Materials

