Adenoviral-Vectored Centralized Consensus Hemagglutinin Vaccine Provides Broad Protection against H2 Influenza a Virus
- PMID: 35746534
- PMCID: PMC9229510
- DOI: 10.3390/vaccines10060926
Adenoviral-Vectored Centralized Consensus Hemagglutinin Vaccine Provides Broad Protection against H2 Influenza a Virus
Abstract
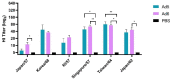
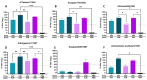
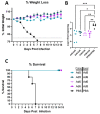
Several influenza pandemics have occurred in the past century, one of which emerged in 1957 from a zoonotic transmission of H2N2 from an avian reservoir into humans. This pandemic caused 2-4 million deaths and circulated until 1968. Since the disappearance of H2N2 from human populations, there has been waning immunity against H2, and this subtype is not currently incorporated into seasonal vaccines. However, H2 influenza remains a pandemic threat due to consistent circulation in avian reservoirs. Here, we describe a method of pandemic preparedness by creating an adenoviral-vectored centralized consensus vaccine design against human H2 influenza. We also assessed the utility of serotype-switching to enhance the protective immune responses seen with homologous prime-boosting strategies. Immunization with an H2 centralized consensus showed a wide breadth of antibody responses after vaccination, protection against challenge with a divergent human H2 strain, and significantly reduced viral load in the lungs after challenge. Further, serotype switching between two species C adenoviruses enhanced protective antibody titers after heterologous boosting. These data support the notion that an adenoviral-vectored H2 centralized consensus vaccine has the ability to provide broadly cross-reactive immune responses to protect against divergent strains of H2 influenza and prepare for a possible pandemic.
Keywords: H2N2; adenovirus; consensus; influenza; species C; vaccine.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Babu T.M., Perera R.A., Wu J.T.K., Fitzgerald T., Nolan C., Cowling B., Krauss S., Treanor J.J., Peiris M. Population Serologic Immunity to Human and Avian H2N2 Viruses in the United States and Hong Kong for Pandemic Risk Assessment. J. Infect. Dis. 2018;218:1054–1060. doi: 10.1093/infdis/jiy291. - DOI - PMC - PubMed
-
- Munster V.J., Baas C., Lexmond P., Waldenstrom J., Wallensten A., Fransson T., Rimmelzwaan G.F., Beyer W.E., Schutten M., Olsen B., et al. Spatial, temporal, and species variation in prevalence of influenza A viruses in wild migratory birds. PLoS Pathog. 2007;3:e61. doi: 10.1371/journal.ppat.0030061. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

