Detam 1 black soybean against cisplatin-induced acute ren failure on rat model via antioxidant, antiinflammatory and antiapoptosis potential
- PMID: 35747350
- PMCID: PMC9209860
- DOI: 10.1016/j.jtcme.2022.01.004
Detam 1 black soybean against cisplatin-induced acute ren failure on rat model via antioxidant, antiinflammatory and antiapoptosis potential
Abstract
Background and aim: Cis-Diamminedichloroplatinum (II) (Cisplatin) is one of the most synthetic anticancer drug but have several adverse effects and one of them is acute ren failure. Cisplatin can induce nephrotoxicity occur via the toxic generation of reactive oxygen species (ROS). Black soybean (Glycine max L. Merr.) has been reported contain high levels of phenolics and anthocyanins that has antioxidant activity. This study aims to determine the effect of ethanol extract of black soybean (EEBS) against cisplatin-induced nephrotoxicity in rats.
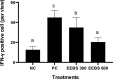
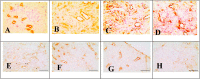
Experimental procedure: Cisplatin-induced nephrotoxicity rats treated with EEBS and the blood samples taken on days 0, 9, and 18. The effects of EEBS was evaluated by determining Interferon-γ (IFN-γ), Caspase-3 (Casp-3), and Interleukin-1β (IL-1β) expression using immunohistochemistry (IHC), blood urea nitrogen (BUN), Uric Acid (UA) content and catalase (CAT) content in the blood plasma with colorimetric assay kit.
Results and conclusion: Based on the results, EEBS treatment had successfully reduced pro-inflammatory cytokines IL-1β and IFN-γ, and improved physiological condition by lowering BUN and UA content while increasing CAT activity. No significant effect was found in Casp-3 expression. EEBS has potential to improve acute renal failure condition through inflammatory suppression and renal function improvement.
Keywords: Blood urea nitrogen; Catalase; Fibronectin; Glycine max L. Merr.; Nephrotoxicity.
© 2022 Center for Food and Biomolecules, National Taiwan University. Production and hosting by Elsevier Taiwan LLC.
Conflict of interest statement
There is no conflict of interest.
Figures









Similar articles
-
Mitigation of ILβ-1, ILβ-6, TNF-α, and markers of apoptosis by ursolic acid against cisplatin-induced oxidative stress and nephrotoxicity in rats.Hum Exp Toxicol. 2021 Dec;40(12_suppl):S397-S405. doi: 10.1177/09603271211045953. Epub 2021 Sep 26. Hum Exp Toxicol. 2021. PMID: 34569348
-
Protective effects of 6-hydroxy-1-methylindole-3-acetonitrile on cisplatin-induced oxidative nephrotoxicity via Nrf2 inactivation.Food Chem Toxicol. 2013 Dec;62:159-66. doi: 10.1016/j.fct.2013.08.039. Epub 2013 Aug 26. Food Chem Toxicol. 2013. PMID: 23989062
-
Carnosic acid attenuates renal injury in an experimental model of rat cisplatin-induced nephrotoxicity.Food Chem Toxicol. 2011 Dec;49(12):3090-7. doi: 10.1016/j.fct.2011.08.018. Epub 2011 Sep 6. Food Chem Toxicol. 2011. PMID: 21930180
-
Anemoside B4 attenuates nephrotoxicity of cisplatin without reducing anti-tumor activity of cisplatin.Phytomedicine. 2019 Mar 15;56:136-146. doi: 10.1016/j.phymed.2018.10.035. Epub 2018 Oct 31. Phytomedicine. 2019. PMID: 30668334
-
Amelioration of cisplatin-induced nephrotoxicity by statins.Indian J Pharmacol. 2013 Jul-Aug;45(4):354-8. doi: 10.4103/0253-7613.115016. Indian J Pharmacol. 2013. PMID: 24014910 Free PMC article.
Cited by
-
Antidiabetic, antidyslipidemia, and renoprotector potency of butterfly pea flower extract (Clitorea ternatea L.) in diabetes mellitus and dyslipidemia rats model.Open Vet J. 2024 May;14(5):1135-1145. doi: 10.5455/OVJ.2024.v14.i5.7. Epub 2024 May 31. Open Vet J. 2024. PMID: 38938424 Free PMC article.
-
Protective effect of yacon leaves extract (Smallanthus sonchifolius (Poepp.) H. Rob) through antifibrosis, anti-inflammatory, and antioxidant mechanisms toward diabetic nephropathy.Res Pharm Sci. 2023 Mar 10;18(3):336-345. doi: 10.4103/1735-5362.371589. eCollection 2023 May-Jun. Res Pharm Sci. 2023. PMID: 37593160 Free PMC article.
-
Cisplatin-Induced Kidney Toxicity: Potential Roles of Major NAD+-Dependent Enzymes and Plant-Derived Natural Products.Biomolecules. 2022 Aug 5;12(8):1078. doi: 10.3390/biom12081078. Biomolecules. 2022. PMID: 36008971 Free PMC article. Review.
-
Antidiabetic and hepatoprotection effect of butterfly pea flower (Clitoria ternatea L.) through antioxidant, anti-inflammatory, lower LDH, ACP, AST, and ALT on diabetes mellitus and dyslipidemia rat.Heliyon. 2024 Apr 16;10(8):e29812. doi: 10.1016/j.heliyon.2024.e29812. eCollection 2024 Apr 30. Heliyon. 2024. PMID: 38681657 Free PMC article.
-
Brazilin cream from Caesalpinia sappan inhibit periodontal disease: in vivo study.PeerJ. 2024 Jul 5;12:e17642. doi: 10.7717/peerj.17642. eCollection 2024. PeerJ. 2024. PMID: 38978754 Free PMC article.
References
-
- Ajith T.A., Usha S., Nivitha V. Ascorbic acid and α-tocopherol protect anticancer drug cisplatin induced nephrotoxicity in mice: a comparative study. Clin Chim Acta. 2007;375:82–86. - PubMed
-
- Manohar S., Leung N. Cisplatin nephrotoxicity: a review of the literature. J Nephrol. 2018;31:15–25. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
