The Response of the Rodent Gut Microbiome to Broad-Spectrum Antibiotics Is Different in Males and Females
- PMID: 35756061
- PMCID: PMC9218673
- DOI: 10.3389/fmicb.2022.897283
The Response of the Rodent Gut Microbiome to Broad-Spectrum Antibiotics Is Different in Males and Females
Abstract
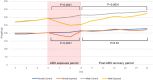
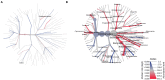
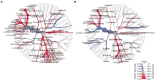
Gut microbiome composition is different in males and females, but sex is rarely considered when prescribing antibiotics, and sex-based differences in gut microbiome recovery following antibiotic treatment are poorly understood. Here, we compared the effects of broad-spectrum antibiotics on both the stool and small bowel microbiomes in male and female rats. Adult male and female Sprague Dawley rats were exposed to a multi-drug antibiotic cocktail for 8 days, or remained unexposed as controls. Following cessation of antibiotics, rats were monitored for an additional 13-day recovery period prior to euthanasia. Baseline stool microbiome composition was similar in males and females. By antibiotic exposure day 8 (AbxD8), exposed male rats exhibited greater loss of stool microbial diversity compared to exposed females, and the relative abundance (RA) of numerous taxa were significantly different in exposed males vs. exposed females. Specifically, RA of phylum Proteobacteria and genera Lactobacillus, Sutterella, Akkermansia, and Serratia were higher in exposed males vs. exposed females, whereas RA of phyla Firmicutes and Actinobacteria and genera Turicibacter and Enterococcus were lower. By 13 days post antibiotics cessation (PAbxD13), the stool RA of these and other taxa remained significantly different from baseline, and also remained significantly different between exposed males and exposed females. RA of phyla Firmicutes and Actinobacteria and genus Enterococcus remained lower in exposed males vs. exposed females, and genus Sutterella remained higher. However, RA of phylum Proteobacteria and genus Akkermansia were now also lower in exposed males vs. females, whereas RA of phylum Bacteroidetes and genus Turicibacter were now higher in exposed males. Further, the small bowel microbiome of exposed rats on PAbxD13 was also significantly different from unexposed controls, with higher RA of Firmicutes, Turicibacter and Parabacteroides in exposed males vs. females, and lower RA of Bacteroidetes, Proteobacteria, Actinobacteria, Oscillospira, Sutterella, and Akkermansia in exposed males vs. females. These findings indicate that broad-spectrum antibiotics have significant and sex-specific effects on gut microbial populations in both stool and the small bowel, and that the recovery of gut microbial populations following exposure to broad-spectrum antibiotics also differs between sexes. These findings may have clinical implications for the way antibiotics are prescribed.
Keywords: antibiotics recovery; antibiotics response; female; male; sex; small bowel microbiome; stool microbiome.
Copyright © 2022 Parodi, Leite, Pimentel, Barlow, Fiorentino, Morales, Pimentel, Weitsman and Mathur.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





References
LinkOut - more resources
Full Text Sources

