Inhibition of autophagy by chloroquine prevents resistance to PI3K/AKT inhibitors and potentiates their antitumor effect in combination with paclitaxel in triple negative breast cancer models
- PMID: 35761360
- PMCID: PMC9235112
- DOI: 10.1186/s12967-022-03462-z
Inhibition of autophagy by chloroquine prevents resistance to PI3K/AKT inhibitors and potentiates their antitumor effect in combination with paclitaxel in triple negative breast cancer models
Abstract
Background: Triple negative breast cancer (TNBC) is an aggressive disease characterized by high risk of relapse and development of resistance to different chemotherapy agents. Several targeted therapies have been investigated in TNBC with modest results in clinical trials. Among these, PI3K/AKT inhibitors have been evaluated in addition to standard therapies, yielding conflicting results and making attempts on elucidating inherent mechanisms of resistance of great interest. Increasing evidences suggest that PI3K/AKT inhibitors can induce autophagy in different cancers. Autophagy represents a supposed mechanism of drug-resistance in aggressive tumors, like TNBC. We, therefore, investigated if two PI3K/AKT inhibitors, ipatasertib and taselisib, could induce autophagy in breast cancer models, and whether chloroquine (CQ), a well known autophagy inhibitor, could potentiate ipatasertib and taselisib anti-cancer effect in combination with conventional chemotherapy.
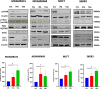
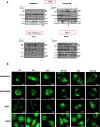
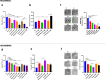
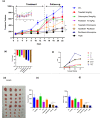
Methods: The induction of autophagy after ipatasertib and taselisib treatment was evaluated in MDAMB231, MDAM468, MCF7, SKBR3 and MDAB361 breast cancer cell lines by assaying LC3-I conversion to LC3-II through immunoblotting and immunofluorescence. Other autophagy-markers as p62/SQSTM1 and ATG5 were evaluated by immunoblotting. Synergistic antiproliferative effect of double and triple combinations of ipatasertib/taselisib plus CQ and/or paclitaxel were evaluated by SRB assay and clonogenic assay. Anti-apoptotic effect of double combination of ipatasertib/taselisib plus CQ was evaluated by increased cleaved-PARP by immunoblot and by Annexin V- flow cytometric analysis. In vivo experiments were performed on xenograft model of MDAMB231 in NOD/SCID mice.
Results: Our results suggested that ipatasertib and taselisib induce increased autophagy signaling in different breast cancer models. This effect was particularly evident in PI3K/AKT resistant TNBC cells, where the inhibition of autophagy by CQ potentiates the therapeutic effect of PI3K/AKT inhibitors in vitro and in vivo TNBC models, synergizing with taxane-based chemotherapy.
Conclusion: These data suggest that inhibition of authophagy with CQ could overcome mechanism of drug resistance to PI3K/AKT inhibitors plus paclitaxel in TNBC making the evaluation of such combinations in clinical trials warranted.
Keywords: Autophagy; Breast Cancer; Chloroquine; PI3K/AKT/mTOR inhibitors; TNBC.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
-
- O’Connor M, Jackman J, Bae I, Myers G. Characterization of the p53 tumor suppressor pathway in cell lines of the national cancer institute anticancer drug screen and correlations with the growth-inhibitory potency of 123 anticancer agents. Cancer Res. 1997;57(19):4285–4300. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

