FBXO38 Ubiquitin Ligase Controls Sertoli Cell Maturation
- PMID: 35769260
- PMCID: PMC9234700
- DOI: 10.3389/fcell.2022.914053
FBXO38 Ubiquitin Ligase Controls Sertoli Cell Maturation
Abstract
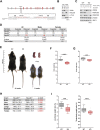
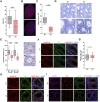
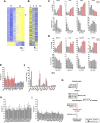
The ubiquitin ligase SCFFBXO38 controls centromeric chromatin by promoting the degradation of the ZXDB protein. To determine the importance of this pathway during development, Fbxo38-deficient mice were generated. The loss of FBXO38 resulted in growth retardation affecting several organs, including the male reproductive system. A detailed analysis of the mutant testes revealed pathological changes in the seminiferous tubules, accompanied by a significant decrease in sperm production and reduced fertility. In adult testes, FBXO38 was specifically expressed in Sertoli cells, a somatic population essential for spermatogenesis initiation and progression. Sertoli cells lacking FBXO38 exhibited stabilized ZXDB protein and upregulated centromeric chromatin. Furthermore, the gene expression profile revealed that the absence of FBXO38 led to a defect in Sertoli cell maturation, specifically characterized by dysregulation in genes controlling retinoic acid metabolism and intercellular communication. Consequently, we documented significant changes in their ability to initiate spermatogonial differentiation. In conclusion, we show that FBXO38 acts as a Sertoli cell maturation factor, affecting the Sertoli cell transcription program, centromere integrity, and, subsequently, the ability to control spermatogenesis.
Keywords: centromere; proteasome; retinoic acid; sertoli cell; spermatogenesis; ubiquitin; ubiquitin ligase.
Copyright © 2022 Dibus, Zobalova, Monleon, Korinek, Filipp, Petrusova, Sedlacek, Kasparek and Cermak.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
FBXO38 Ubiquitin Ligase Controls Centromere Integrity via ZXDA/B Stability.Front Cell Dev Biol. 2022 Jun 23;10:929288. doi: 10.3389/fcell.2022.929288. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35813202 Free PMC article.
-
FBXO38 is dispensable for PD-1 regulation.EMBO Rep. 2024 Oct;25(10):4206-4225. doi: 10.1038/s44319-024-00220-8. Epub 2024 Sep 12. EMBO Rep. 2024. PMID: 39266770 Free PMC article.
-
Establishment, maintenance and functional integrity of the blood-testis barrier in organotypic cultures of fresh and frozen/thawed prepubertal mouse testes.Mol Hum Reprod. 2017 May 1;23(5):304-320. doi: 10.1093/molehr/gax017. Mol Hum Reprod. 2017. PMID: 28333312
-
Molecular Mechanisms and Signaling Pathways Involved in Sertoli Cell Proliferation.Front Endocrinol (Lausanne). 2019 Apr 16;10:224. doi: 10.3389/fendo.2019.00224. eCollection 2019. Front Endocrinol (Lausanne). 2019. PMID: 31040821 Free PMC article. Review.
-
Somatic cell-germ cell relationships in mammalian testes during development and spermatogenesis.Ciba Found Symp. 1994;182:271-4; discussion 274-81. doi: 10.1002/9780470514573.ch15. Ciba Found Symp. 1994. PMID: 7835155 Review.
Cited by
-
Molecular function of Krüppel-like factor 7 in biology.Acta Biochim Biophys Sin (Shanghai). 2023 May 24;55(5):713-725. doi: 10.3724/abbs.2023061. Acta Biochim Biophys Sin (Shanghai). 2023. PMID: 37227154 Free PMC article. Review.
-
FBXO38 Ubiquitin Ligase Controls Centromere Integrity via ZXDA/B Stability.Front Cell Dev Biol. 2022 Jun 23;10:929288. doi: 10.3389/fcell.2022.929288. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35813202 Free PMC article.
-
The emerging and diverse roles of F-box proteins in spermatogenesis and male infertility.Cell Regen. 2024 Jun 26;13(1):13. doi: 10.1186/s13619-024-00196-9. Cell Regen. 2024. PMID: 38918264 Free PMC article. Review.
-
FBXO38 is dispensable for PD-1 regulation.EMBO Rep. 2024 Oct;25(10):4206-4225. doi: 10.1038/s44319-024-00220-8. Epub 2024 Sep 12. EMBO Rep. 2024. PMID: 39266770 Free PMC article.
-
Cytoplasmic FBXO38 mediates PD-1 degradation.EMBO Rep. 2024 Oct;25(10):4168-4171. doi: 10.1038/s44319-024-00254-y. Epub 2024 Sep 16. EMBO Rep. 2024. PMID: 39294504 Free PMC article.
References
-
- Auharek S. A., de França L. R. (2010). Postnatal Testis Development, Sertoli Cell Proliferation and Number of Different Spermatogonial Types in C57BL/6J Mice Made Transiently Hypo- and Hyperthyroidic during the Neonatal Period. J. Anat. 216 (5), 577–588. 10.1111/j.1469-7580.2010.01219.x - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

