Metformin, Rapamycin, or Nicotinamide Mononucleotide Pretreatment Attenuate Cognitive Impairment After Cerebral Hypoperfusion by Inhibiting Microglial Phagocytosis
- PMID: 35769369
- PMCID: PMC9234123
- DOI: 10.3389/fneur.2022.903565
Metformin, Rapamycin, or Nicotinamide Mononucleotide Pretreatment Attenuate Cognitive Impairment After Cerebral Hypoperfusion by Inhibiting Microglial Phagocytosis
Abstract
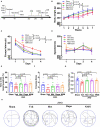
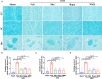
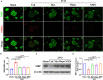
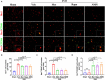
Vascular cognitive impairment (VCI) is the second leading form of dementia after Alzheimer's disease (AD) plaguing the elder population. Despite the enormous prevalence of VCI, the biological basis of this disease has been much less well-studied than that of AD, with no specific therapy currently existing to prevent or treat VCI. As VCI mainly occurs in the elderly, the role of anti-aging drugs including metformin, rapamycin, and nicotinamide mono nucleotide (NMN), and the underlying mechanism remain uncertain. Here, we examined the role of metformin, rapamycin, and NMN in cognitive function, white matter integrity, microglial response, and phagocytosis in a rat model of VCI by bilateral common carotid artery occlusion (BCCAO). BCCAO-induced chronic cerebral hypoperfusion could cause spatial working memory deficits and white matter lesions (WMLs), along with increasing microglial activation and phagocytosis compared to sham-operated rats. We found the cognitive impairment was significantly improved in BCCAO rats pretreated with these three drugs for 14 days before BCCAO compared with the vehicle group by the analysis of the Morris water maze and new object recognition tests. Pretreatment of metformin, rapamycin, or NMN also increased myelin basic protein (MBP, a marker for myelin) expression and reduced SMI32 (a marker for demyelinated axons) intensity and SMI32/MBP ratio compared with the vehicle group, suggesting that these drugs could ameliorate BCCAO-induced WMLs. The findings were confirmed by Luxol fast blue (LFB) stain, which is designed for staining myelin/myelinated axons. We further found that pretreatment of metformin, rapamycin, or NMN reduced microglial activation and the number of M1 microglia, but increased the number of M2 microglia compared to the vehicle group. Importantly, the number of MBP+/Iba1+/CD68+ microglia was significantly reduced in the BCCAO rats pretreated with these three drugs compared with the vehicle group, suggesting that these drugs suppress microglial phagocytosis. No significant difference was found between the groups pretreated with metformin, rapamycin, or NMN. Our data suggest that metformin, rapamycin, or NMN could protect or attenuate cognitive impairment and WMLs by modifying microglial polarization and inhibiting phagocytosis. The findings may open a new avenue for VCI treatment.
Keywords: metformin; microglia; nicotinamide mono-nucleotide; rapamycin; vascular cognitive impairment.
Copyright © 2022 Yu, Zheng, Cheng, Shao, Zhuge and Jin.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
The Combination of Citicoline and Nicotinamide Mononucleotide Induces Neurite Outgrowth and Mitigates Vascular Cognitive Impairment via SIRT1/CREB Pathway.Cell Mol Neurobiol. 2023 Nov;43(8):4261-4277. doi: 10.1007/s10571-023-01416-7. Epub 2023 Oct 9. Cell Mol Neurobiol. 2023. PMID: 37812361 Free PMC article.
-
Hyperforin ameliorates neuroinflammation and white matter lesions by regulating microglial VEGFR2 /SRC pathway in vascular cognitive impairment mice.CNS Neurosci Ther. 2024 Mar;30(3):e14666. doi: 10.1111/cns.14666. CNS Neurosci Ther. 2024. PMID: 38468126 Free PMC article.
-
Berberine Ameliorates Cognitive Impairment by Regulating Microglial Polarization and Increasing Expression of Anti-inflammatory Factors following Permanent Bilateral Common Carotid Artery Occlusion in Rats.CNS Neurol Disord Drug Targets. 2022;21(9):869-879. doi: 10.2174/1871527321666220124140323. CNS Neurol Disord Drug Targets. 2022. PMID: 35142272
-
Animal Models of Chronic Cerebral Hypoperfusion: From Mouse to Primate.Int J Mol Sci. 2019 Dec 7;20(24):6176. doi: 10.3390/ijms20246176. Int J Mol Sci. 2019. PMID: 31817864 Free PMC article. Review.
-
Intermittent Metabolic Switching and Vascular Cognitive Impairment.J Obes Metab Syndr. 2024 Jun 30;33(2):92-107. doi: 10.7570/jomes24010. Epub 2024 May 13. J Obes Metab Syndr. 2024. PMID: 38736362 Free PMC article. Review.
Cited by
-
The identification of new roles for nicotinamide mononucleotide after spinal cord injury in mice: an RNA-seq and global gene expression study.Front Cell Neurosci. 2023 Dec 14;17:1323566. doi: 10.3389/fncel.2023.1323566. eCollection 2023. Front Cell Neurosci. 2023. PMID: 38155866 Free PMC article.
-
The Combination of Citicoline and Nicotinamide Mononucleotide Induces Neurite Outgrowth and Mitigates Vascular Cognitive Impairment via SIRT1/CREB Pathway.Cell Mol Neurobiol. 2023 Nov;43(8):4261-4277. doi: 10.1007/s10571-023-01416-7. Epub 2023 Oct 9. Cell Mol Neurobiol. 2023. PMID: 37812361 Free PMC article.
-
Single-cell RNA Sequencing Identifies a Novel Subtype of Microglia with High Cd74 Expression that Facilitates White Matter Inflammation During Chronic Cerebral Hypoperfusion.Neurochem Res. 2024 Oct;49(10):2821-2841. doi: 10.1007/s11064-024-04206-9. Epub 2024 Jul 16. Neurochem Res. 2024. PMID: 39012534
-
Unraveling the complex pathophysiology of white matter hemorrhage in intracerebral stroke: A single-cell RNA sequencing approach.CNS Neurosci Ther. 2024 Mar;30(3):e14652. doi: 10.1111/cns.14652. CNS Neurosci Ther. 2024. PMID: 38433011 Free PMC article.
-
The evolving pathophysiology of TBI and the advantages of temporally-guided combination therapies.Neurochem Int. 2024 Nov;180:105874. doi: 10.1016/j.neuint.2024.105874. Epub 2024 Oct 2. Neurochem Int. 2024. PMID: 39366429 Review.
References
-
- Gorelick PB, Scuteri A, Black SE, DeCarli C, Greenberg SM, Iadecola C, et al. . Vascular contributions to cognitive impairment and dementia. a statement for healthcare professionals from the american heart association/american stroke association. Stroke. (2011) 42:2672–713. 10.1161/STR.0b013e3182299496 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

