Pro-Neurotensin as a Potential Novel Diagnostic Biomarker for Detection of Nonalcoholic Fatty Liver Disease
- PMID: 35769889
- PMCID: PMC9234179
- DOI: 10.2147/DMSO.S365147
Pro-Neurotensin as a Potential Novel Diagnostic Biomarker for Detection of Nonalcoholic Fatty Liver Disease
Abstract
Background and aims: Currently, liver biopsy is the gold standard method for diagnosis of non-alcoholic fatty liver severity. It is critical to develop non-invasive diagnostic method to diagnose nonalcoholic fatty liver rather than invasive techniques. Our case-control study was to address the value of circulating miRNA-122 and serum pro-neurotensin as a potential non-invasive biomarker for the diagnosis of non-alcoholic fatty acid diseases.
Methods: Clinical assessment, laboratory investigations, and anthropometric measurements were reported for 157 patients with proven NAFLD. Apparently, healthy participants (n=100) were enrolled as a control group. Serum samples were tested for micro-RNAs-122 and pro-neurotensin.
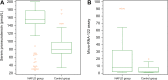
Results: Compared with the control subjects, both mi-RNA-122 and serum proneurotensin levels were increased in NAFLD (p<0.001) and at a cut-off ≥6.83, mi-RNA-122 had 51.0% sensitivity, 70.0% specificity to differentiate NAFLD from healthy controls, while serum proneurotensin had 80.0% sensitivity and 80.0% specificity at a cutoff ≥108.
Conclusion: The circulating pro-neurotensin might be used as a novel biomarker for diagnosis of patients with NAFLD, wherefore the integration of a circulating mi-RNA-122 and serum pro-neurotensin could be beneficial to diagnose NAFLD cases. Large-scale studies are needed to investigate the possible role of mi-RNA-122 and pro-neurotensin in the development, progression, and prognosis of NAFLD and NASH.
Keywords: MAFLD; NAFLD; biomarker; diagnosis; fatty liver; metabolic associated fatty liver disease; micro-RNA-122; nonalcoholic fatty liver disease; noninvasive; proneurotensin.
© 2022 Mohamed et al.
Conflict of interest statement
The authors declare no conflict of interest, financial or otherwise.
Figures
References
LinkOut - more resources
Full Text Sources
Medical