Mechanisms of membrane protein crystallization in 'bicelles'
- PMID: 35773455
- PMCID: PMC9246360
- DOI: 10.1038/s41598-022-13945-0
Mechanisms of membrane protein crystallization in 'bicelles'
Abstract
Despite remarkable progress, mainly due to the development of LCP and 'bicelle' crystallization, lack of structural information remains a bottleneck in membrane protein (MP) research. A major reason is the absence of complete understanding of the mechanism of crystallization. Here we present small-angle scattering studies of the evolution of the "bicelle" crystallization matrix in the course of MP crystal growth. Initially, the matrix corresponds to liquid-like bicelle state. However, after adding the precipitant, the crystallization matrix transforms to jelly-like state. The data suggest that this final phase is composed of interconnected ribbon-like bilayers, where crystals grow. A small amount of multilamellar phase appears, and its volume increases concomitantly with the volume of growing crystals. We suggest that the lamellar phase surrounds the crystals and is critical for crystal growth, which is also common for LCP crystallization. The study discloses mechanisms of "bicelle" MP crystallization and will support rational design of crystallization.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
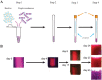

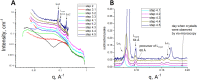
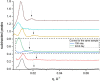

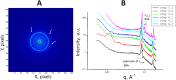


References
-
- Yeagle PL. The Membranes of Cells (Third Edition) Academic Press; 2016. Chapter 10—Membrane Proteins; pp. 219–268.
-
- MPSTRUC. Membrane Proteins of Known 3D Structure. https://blanco.biomol.uci.edu/mpstruc/.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

