Analysis of chemomechanical behavior of stress fibers by continuum mechanics-based FRAP
- PMID: 35778840
- PMCID: PMC9388576
- DOI: 10.1016/j.bpj.2022.06.032
Analysis of chemomechanical behavior of stress fibers by continuum mechanics-based FRAP
Abstract
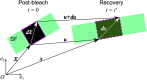
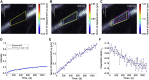
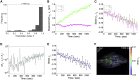
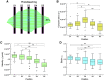
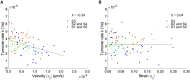
Fluorescence recovery after photobleaching (FRAP) is a common technique to analyze the turnover of molecules in living cells. Numerous physicochemical models have been developed to quantitatively evaluate the rate of turnover driven by chemical reaction and diffusion that occurs in a few seconds to minutes. On the other hand, they have limitations in interpreting long-term FRAP responses where intracellular active movement inevitably provides target molecular architectures with additional effects other than chemical reaction and diffusion, namely directed transport and structural deformation. To overcome the limitations, we develop a continuum mechanics-based model that allows for decoupling FRAP response into the intrinsic turnover rate and subcellular mechanical characteristics such as displacement vector and strain tensor. Our approach was validated using fluorescently labeled β-actin in an actomyosin-mediated contractile apparatus called stress fibers, revealing spatially distinct patterns of the multi-physicochemical events, in which the turnover rate, which represents effective off-rate of β-actin, was significantly higher at the center of the cell. We also found that the turnover rate is negatively correlated with the rate of displacement or velocity along stress fibers but, interestingly, not with the absolute magnitude of strain. Moreover, stress fibers are subjected to centripetal flow that is facilitated by the circulation of actin molecules. Taken together, this novel framework for long-term FRAP analysis allows for unveiling the contribution of overlooked microscopic mechanics to molecular turnover in living cells.
Copyright © 2022 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures


Similar articles
-
Long-term molecular turnover of actin stress fibers revealed by advection-reaction analysis in fluorescence recovery after photobleaching.PLoS One. 2022 Nov 7;17(11):e0276909. doi: 10.1371/journal.pone.0276909. eCollection 2022. PLoS One. 2022. PMID: 36342915 Free PMC article.
-
Long-Term Fluorescence Recovery After Photobleaching (FRAP).Methods Mol Biol. 2023;2600:311-322. doi: 10.1007/978-1-0716-2851-5_21. Methods Mol Biol. 2023. PMID: 36587107
-
CM-FRAP-Continuum Mechanics-Based Fluorescence Recovery After Photobleaching.Curr Protoc. 2023 Jan;3(1):e655. doi: 10.1002/cpz1.655. Curr Protoc. 2023. PMID: 36689324
-
Fluorescence recovery after photobleaching: application to nuclear proteins.Adv Biochem Eng Biotechnol. 2005;95:177-99. doi: 10.1007/b102214. Adv Biochem Eng Biotechnol. 2005. PMID: 16080269 Review.
-
Mechanostress resistance involving formin homology proteins: G- and F-actin homeostasis-driven filament nucleation and helical polymerization-mediated actin polymer stabilization.Biochem Biophys Res Commun. 2018 Nov 25;506(2):323-329. doi: 10.1016/j.bbrc.2018.09.189. Epub 2018 Oct 9. Biochem Biophys Res Commun. 2018. PMID: 30309655 Review.
Cited by
-
Intracellular Macromolecular Crowding within Individual Stress Fibers Analyzed by Fluorescence Correlation Spectroscopy.Cell Mol Bioeng. 2024 Jun 7;17(3):165-176. doi: 10.1007/s12195-024-00803-4. eCollection 2024 Jun. Cell Mol Bioeng. 2024. PMID: 39050511 Free PMC article.
-
Long-term molecular turnover of actin stress fibers revealed by advection-reaction analysis in fluorescence recovery after photobleaching.PLoS One. 2022 Nov 7;17(11):e0276909. doi: 10.1371/journal.pone.0276909. eCollection 2022. PLoS One. 2022. PMID: 36342915 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

