The LncRNA AK018453 regulates TRAP1/Smad signaling in IL-17-activated astrocytes: A potential role in EAE pathogenesis
- PMID: 35778934
- PMCID: PMC9545958
- DOI: 10.1002/glia.24239
The LncRNA AK018453 regulates TRAP1/Smad signaling in IL-17-activated astrocytes: A potential role in EAE pathogenesis
Abstract
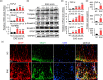
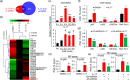
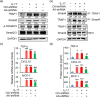
The pro-inflammatory cytokine interleukin 17 (IL-17), that is mainly produced by Th17 cells, has been recognized as a key regulator in multiple sclerosis (MS) and experimental autoimmune encephalomyelitis (EAE). Reactive astrocytes stimulated by proinflammatory cytokines including IL-17 are involved in blood brain barrier destruction, inflammatory cells infiltration and spinal cord injury. However, the role of long non-coding RNAs (lncRNAs) induced by IL-17 in the pathogenesis of MS and EAE remains unknown. Herein, we found that an IL-17-induced lncRNA AK018453 promoted TGF-β receptor-associated protein 1 (TRAP1) expression and Smad-dependent signaling in mouse primary astrocytes. Knockdown of AK018453 significantly suppressed astrocytosis, attenuated the phosphorylation of Smad2/3, reduced NF-κB p65 and CBP/P300 binding to the TRAP1 promoter, and diminished pro-inflammatory cytokine production in the IL-17-treated astrocytes. AK018453 knockdown in astrocytes by a lentiviral vector in vivo dramatically inhibited inflammation and prevented the mice from demyelination in the spinal cord during the progression of EAE. Together, these results suggest that AK018453 regulates IL-17-dependent inflammatory response in reactive astrocytes and potentially promotes the pathogenesis of EAE via the TRAP1/Smad pathway. Targeting this pathway may have a therapeutic potential for intervening inflammatory demyelinating diseases.
Keywords: TGF-β receptor-associated protein 1; astrocytes; experimental autoimmune encephalomyelitis; lncRNA AK018453; multiple sclerosis.
© 2022 The Authors. GLIA published by Wiley Periodicals LLC.
Conflict of interest statement
The authors declare no potential conflict of interest.
Figures





Similar articles
-
IL-9-triggered lncRNA Gm13568 regulates Notch1 in astrocytes through interaction with CBP/P300: contribute to the pathogenesis of experimental autoimmune encephalomyelitis.J Neuroinflammation. 2021 May 11;18(1):108. doi: 10.1186/s12974-021-02156-5. J Neuroinflammation. 2021. PMID: 33971906 Free PMC article.
-
Interleukin-17 (IL-17)-induced microRNA 873 (miR-873) contributes to the pathogenesis of experimental autoimmune encephalomyelitis by targeting A20 ubiquitin-editing enzyme.J Biol Chem. 2014 Oct 17;289(42):28971-86. doi: 10.1074/jbc.M114.577429. Epub 2014 Sep 2. J Biol Chem. 2014. PMID: 25183005 Free PMC article.
-
Analysis of Long Noncoding RNA and mRNA Expression Profiles in IL-9-Activated Astrocytes and EAE Mice.Cell Physiol Biochem. 2018;45(5):1986-1998. doi: 10.1159/000487975. Epub 2018 Mar 2. Cell Physiol Biochem. 2018. PMID: 29518786
-
MiR-409-3p and MiR-1896 co-operatively participate in IL-17-induced inflammatory cytokine production in astrocytes and pathogenesis of EAE mice via targeting SOCS3/STAT3 signaling.Glia. 2019 Jan;67(1):101-112. doi: 10.1002/glia.23530. Epub 2018 Oct 7. Glia. 2019. PMID: 30294880
-
Role of Th17 cells in the pathogenesis of CNS inflammatory demyelination.J Neurol Sci. 2013 Oct 15;333(1-2):76-87. doi: 10.1016/j.jns.2013.03.002. Epub 2013 Apr 8. J Neurol Sci. 2013. PMID: 23578791 Free PMC article. Review.
Cited by
-
Multiple Sclerosis: Roles of miRNA, lcnRNA, and circRNA and Their Implications in Cellular Pathways.Int J Mol Sci. 2024 Feb 13;25(4):2255. doi: 10.3390/ijms25042255. Int J Mol Sci. 2024. PMID: 38396932 Free PMC article. Review.
-
Assessing the suitability of long non-coding RNAs as therapeutic targets and biomarkers in SARS-CoV-2 infection.Front Mol Biosci. 2022 Aug 16;9:975322. doi: 10.3389/fmolb.2022.975322. eCollection 2022. Front Mol Biosci. 2022. PMID: 36052163 Free PMC article. Review.
-
Interaction between Th17 and central nervous system in multiple sclerosis.Brain Behav Immun Health. 2024 Dec 24;43:100928. doi: 10.1016/j.bbih.2024.100928. eCollection 2025 Feb. Brain Behav Immun Health. 2024. PMID: 39845807 Free PMC article. Review.
-
Epigenetic and Mitochondrial Metabolic Dysfunction in Multiple Sclerosis: A Review of Herbal Drug Approaches and Current Clinical Trials.Mol Neurobiol. 2025 Aug;62(8):10045-10090. doi: 10.1007/s12035-025-04868-8. Epub 2025 Apr 3. Mol Neurobiol. 2025. PMID: 40180689 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

