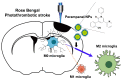
Perampanel Reduces Brain Damage via Induction of M2 Microglia in a Neonatal Rat Stroke Model
- PMID: 35782016
- PMCID: PMC9248959
- DOI: 10.2147/IJN.S361377
Perampanel Reduces Brain Damage via Induction of M2 Microglia in a Neonatal Rat Stroke Model
Abstract
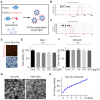
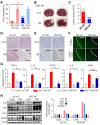
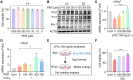
Purpose: Ischemic stroke is a leading cause of death and disability worldwide. Additionally, neonatal ischemia is a common cause of neonatal brain injury, resulting in cerebral palsy with subsequent learning disabilities and epilepsy. However, there is currently a lack of effective treatments available for patients with perinatal ischemic stroke. In this study, we investigated the effect of perampanel (PER)-loaded poly lactic-co-glycolic acid (PLGA) by targeting microglia in perinatal stroke.
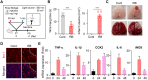
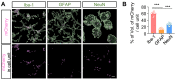
Methods: After formation of focal ischemic stroke by photothrombosis in P7 rats, PER-loaded PLGA was injected intrathecally. Proinflammatory markers (TNF-α, IL-1β, IL-6, COX2, and iNOS) and M2 polarization markers (Ym1 and Arg1) were evaluated. We investigated whether PER increased M2 microglial polarization in vitro.
Results: PER-loaded PLGA nanoparticles decreased the pro-inflammatory cytokines compared to the control group. Furthermore, they increased M2 polarization.
Conclusion: PER-loaded PLGA nanoparticles decreased the size of the infarct and increased motor function in a perinatal ischemic stroke rat model. Pro-inflammatory cytokines were also reduced compared to the control group. Finally, this development of a drug delivery system targeting microglia confirms the potential to develop new therapeutic agents for perinatal ischemic stroke.
Keywords: PLGA; ischemic stroke; microglial polarization; nanoparticle; neonate; perampanel; poly lactic-co-glycolic acid.
© 2022 Shin et al.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures
Similar articles
-
The Recent Applications of PLGA-Based Nanostructures for Ischemic Stroke.Pharmaceutics. 2023 Sep 14;15(9):2322. doi: 10.3390/pharmaceutics15092322. Pharmaceutics. 2023. PMID: 37765291 Free PMC article. Review.
-
Deficiency in the voltage-gated proton channel Hv1 increases M2 polarization of microglia and attenuates brain damage from photothrombotic ischemic stroke.J Neurochem. 2016 Oct;139(1):96-105. doi: 10.1111/jnc.13751. Epub 2016 Sep 9. J Neurochem. 2016. PMID: 27470181 Free PMC article.
-
Lipoxin A4 regulates microglial M1/M2 polarization after cerebral ischemia-reperfusion injury via the Notch signaling pathway.Exp Neurol. 2021 May;339:113645. doi: 10.1016/j.expneurol.2021.113645. Epub 2021 Feb 15. Exp Neurol. 2021. PMID: 33600815
-
PINK1 siRNA-loaded poly(lactic-co-glycolic acid) nanoparticles provide neuroprotection in a mouse model of photothrombosis-induced ischemic stroke.Glia. 2023 May;71(5):1294-1310. doi: 10.1002/glia.24339. Epub 2023 Jan 18. Glia. 2023. PMID: 36655313
-
The natural (poly)phenols as modulators of microglia polarization via TLR4/NF-κB pathway exert anti-inflammatory activity in ischemic stroke.Eur J Pharmacol. 2022 Jan 5;914:174660. doi: 10.1016/j.ejphar.2021.174660. Epub 2021 Dec 1. Eur J Pharmacol. 2022. PMID: 34863710 Review.
Cited by
-
The inflammasome-activating poxvirus peptide IAMP29 promotes antimicrobial and anticancer responses.Exp Mol Med. 2024 Nov;56(11):2475-2490. doi: 10.1038/s12276-024-01339-3. Epub 2024 Nov 1. Exp Mol Med. 2024. PMID: 39511430 Free PMC article.
-
Perioperative perampanel administration for early seizure prophylaxis in brain tumor patients.Surg Neurol Int. 2023 Aug 11;14:287. doi: 10.25259/SNI_495_2023. eCollection 2023. Surg Neurol Int. 2023. PMID: 37680915 Free PMC article.
-
The Recent Applications of PLGA-Based Nanostructures for Ischemic Stroke.Pharmaceutics. 2023 Sep 14;15(9):2322. doi: 10.3390/pharmaceutics15092322. Pharmaceutics. 2023. PMID: 37765291 Free PMC article. Review.
-
Nanomaterial payload delivery to central nervous system glia for neural protection and repair.Front Cell Neurosci. 2023 Oct 24;17:1266019. doi: 10.3389/fncel.2023.1266019. eCollection 2023. Front Cell Neurosci. 2023. PMID: 37941607 Free PMC article. Review.
-
Rejuvenating aged microglia by p16ink4a-siRNA-loaded nanoparticles increases amyloid-β clearance in animal models of Alzheimer's disease.Mol Neurodegener. 2024 Mar 16;19(1):25. doi: 10.1186/s13024-024-00715-x. Mol Neurodegener. 2024. PMID: 38493185 Free PMC article.
References
-
- Ferriero DM, Fullerton HJ, Bernard TJ, et al. Management of stroke in neonates and children: a scientific statement from the American association/American Stroke Association. Stroke. 2019;50(3):e51–e96. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

