Therapeutic Efficacy of Carvacrol-Loaded Nanoemulsion in a Mouse Model of Schistosomiasis
- PMID: 35784725
- PMCID: PMC9247328
- DOI: 10.3389/fphar.2022.917363
Therapeutic Efficacy of Carvacrol-Loaded Nanoemulsion in a Mouse Model of Schistosomiasis
Abstract
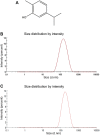
Since praziquantel is the only drug available to treat schistosomiasis, a neglected parasitic disease that affects more than 240 million people worldwide, there is an urgent demand for new antischistosomal agents. Natural compound-loaded nanoparticles have recently emerged as a promising alternative for the treatment of schistosomiasis. Carvacrol is an antimicrobial monoterpene present in the essential oil extracted from several plants, especially oregano (Origanum vulgare). In this study, a carvacrol nanoemulsion (CVNE) was prepared, characterized, and administered orally (200 mg/kg) in a mouse infected with either immature (prepatent infection) or adult (patent infection) Schistosoma mansoni. For comparison, data obtained with an unloaded nanoemulsion (blank formulation), free carvacrol, and the drug of reference praziquantel are also presented. CVNE was more effective than free carvacrol in reducing the worm burden and egg production in both patent and prepatent infections. Favorably, CVNE had a high effect in terms of reducing the number of worms and eggs (85%-90%) compared with praziquantel (∼30%) in prepatent infection. In tandem, carvacrol-loaded nanoemulsion markedly improved antischistosomal activity, showing efficiency in reducing worm and egg burden, and thus it may be a promising delivery system for the treatment of schistosomiasis.
Keywords: antischistosomal compounds; drug delivery; infectious diseases; nanoemulsion; nanotechnology; natural product.
Copyright © 2022 Xavier, de Souza, Rodrigues, Melo, Roquini, Lemes, Wilairatana, Oliveira and de Moraes.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Amorim C. R., Pavani T. F. A., Lopes A. F. S., Duque M. D., Mengarda A. C. A., Silva M. P., et al. (2020). Schiff Bases of 4-Phenyl-2-Aminothiazoles as Hits to New Antischistosomals: Synthesis, In Vitro, In Vivo and In Silico Studies. Eur. J. Pharm. Sci. 150, 105371. 10.1016/j.ejps.2020.105371 - DOI - PubMed
LinkOut - more resources
Full Text Sources

