Sensitivity of portable low-field magnetic resonance imaging for multiple sclerosis lesions
- PMID: 35792417
- PMCID: PMC9421456
- DOI: 10.1016/j.nicl.2022.103101
Sensitivity of portable low-field magnetic resonance imaging for multiple sclerosis lesions
Abstract
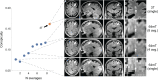
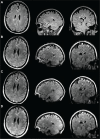
Magnetic resonance imaging (MRI) is a fundamental tool in the diagnosis and management of neurological diseases such as multiple sclerosis (MS). New portable, low-field strength, MRI scanners could potentially lower financial and technical barriers to neuroimaging and reach underserved or disabled populations, but the sensitivity of these devices for MS lesions is unknown. We sought to determine if white matter lesions can be detected on a portable 64mT scanner, compare automated lesion segmentations and total lesion volume between paired 3T and 64mT scans, identify features that contribute to lesion detection accuracy, and explore super-resolution imaging at low-field. In this prospective, cross-sectional study, same-day brain MRI (FLAIR, T1w, and T2w) scans were collected from 36 adults (32 women; mean age, 50 ± 14 years) with known or suspected MS using Siemens 3T (FLAIR: 1 mm isotropic, T1w: 1 mm isotropic, and T2w: 0.34-0.5 × 0.34-0.5 × 3-5 mm) and Hyperfine 64mT (FLAIR: 1.6 × 1.6 × 5 mm, T1w: 1.5 × 1.5 × 5 mm, and T2w: 1.5 × 1.5 × 5 mm) scanners at two centers. Images were reviewed by neuroradiologists. MS lesions were measured manually and segmented using an automated algorithm. Statistical analyses assessed accuracy and variability of segmentations across scanners and systematic scanner biases in automated volumetric measurements. Lesions were identified on 64mT scans in 94% (31/33) of patients with confirmed MS. The average smallest lesions manually detected were 5.7 ± 1.3 mm in maximum diameter at 64mT vs 2.1 ± 0.6 mm at 3T, approaching the spatial resolution of the respective scanner sequences (3T: 1 mm, 64mT: 5 mm slice thickness). Automated lesion volume estimates were highly correlated between 3T and 64mT scans (r = 0.89, p < 0.001). Bland-Altman analysis identified bias in 64mT segmentations (mean = 1.6 ml, standard error = 5.2 ml, limits of agreement = -19.0-15.9 ml), which over-estimated low lesion volume and under-estimated high volume (r = 0.74, p < 0.001). Visual inspection revealed over-segmentation was driven venous hyperintensities on 64mT T2-FLAIR. Lesion size drove segmentation accuracy, with 93% of lesions > 1.0 ml and all lesions > 1.5 ml being detected. Using multi-acquisition volume averaging, we were able to generate 1.6 mm isotropic images on the 64mT device. Overall, our results demonstrate that in established MS, a portable 64mT MRI scanner can identify white matter lesions, and that automated estimates of total lesion volume correlate with measurements from 3T scans.
Keywords: Hyperfine; Low-field MRI; Multiple sclerosis; Point-of-care MRI; Portable MRI; White matter lesions.
Copyright © 2022 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Joel M. Stein reports financial support was provided by Hyperfine. Daniel S. Reich reports financial support was provided by Abata Therapeutics. Daniel S. Reich reports financial support was provided by Sanofi Genzyme. Daniel S. Reich reports financial support was provided by Vertex Pharmaceuticals. Samantha By reports a relationship with Hyperfine that includes: employment. Samantha By reports a relationship with Bristol Myers Squibb Co that includes: employment. Russell T. Shinohara reports a relationship with Octave Bioscience that includes: consulting or advisory. Russell T. Shinohara reports a relationship with American Medical Association that includes: consulting or advisory. Joel M. Stein reports a relationship with Centaur Diagnostics that includes: consulting or advisory.
Figures







References
-
- Avants B.B., Tustison N., Song G. Advanced normalization tools (ANTS) Insight j. 2009;2:1–35.
-
- Bendszus M., Roberts D., Kolumban B., Meza J.A., Bereczki D., San-Juan D., Liu B.P., Anzalone N., Maravilla K. Dose Finding Study of Gadopiclenol, a New Macrocyclic Contrast Agent, in MRI of Central Nervous System. Invest. Radiol. Lippincott Williams and Wilkins. 2020;55(3):129–137. - PubMed
-
- Campbell-Washburn A.E., Ramasawmy R., Restivo M.C., Bhattacharya I., Basar B., Herzka D.A., Hansen M.S., Rogers T., Bandettini W.P., McGuirt D.R., Mancini C., Grodzki D., Schneider R., Majeed W., Bhat H., Xue H., Moss J., Malayeri A.A., Jones E.C., Koretsky A.P., Kellman P., Chen M.Y., Lederman R.J., Balaban R.S. Opportunities in Interventional and Diagnostic Imaging by Using High-Performance Low-Field-Strength MRI. Radiology. 2019;293(2):384–393. - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

