Humoral immune response to SARS-CoV-2 third vaccination in patients with multiple sclerosis and healthy controls: A prospective multicenter study
- PMID: 35797803
- PMCID: PMC9250418
- DOI: 10.1016/j.msard.2022.104009
Humoral immune response to SARS-CoV-2 third vaccination in patients with multiple sclerosis and healthy controls: A prospective multicenter study
Abstract
Background: Third vaccination against SARS-CoV-2 is recommended for patients with multiple sclerosis (pwMS), usually six months after the last vaccination.
Methods: In this prospective multicenter study on 292 pwMS and 46 healthy controls (HC), who had all received two vaccinations prior to study enrollment, SARS-CoV-2 IgG response was measured in the month before and 2-4 months after third vaccination. PwMS were categorized as follows: untreated (N-DMT, n = 32), receiving disease-modifying therapy (DMT) with expected humoral response (er-DMT: interferon-beta preparations, glatiramer acetate, dimethyl fumarate, teriflunomide, natalizumab, cladribine, alemtuzumab; n = 120) or no expected humoral response (nr-DMT: S1PMs, CD20mAb; n = 140).
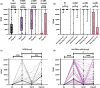
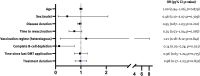
Results: PwMS on nr-DMT had significantly lower median antibody levels before (12.1 U/ml [0.4-2500]) and after third vaccination (305 U/ml [0.4-2500]) in comparison to other groups (p<0.001). We did not find differences in antibody levels after homologous (n = 281; 2500 [0.4-2500]) and heterologous (n = 57; 2500 [0.4-2500]) vaccination regime regardless of the DMT group. The DMT group (β= -0.60; 95% CI -1195.73, -799.10; p<0.001) was associated with antibody levels after third vaccination, while time to revaccination (6 months [1-13]) was not. After third vaccination, seropositivity was reached in 75.8% and 82.2% of pwMS on anti-CD20 mAbs and S1PMs, respectively. Complete B-cell depletion significantly decreased the probability of seroconversion even after the third vaccination (OR 0.14; p = 0.021), whereas time interval to last DMT intake and time to revaccination did not. Twenty-two patients reported a SARS-CoV-2 infection (3 N-DMT, 9 er-DMT, 10 nr-DMT), one being asymptomatic and the rest having a mild course.
Conclusion: Humoral response to SARS-CoV-2 third vaccination in pwMS is excellent. While reduced by S1PMs and CD20mAb, protective response is still expected in the majority of patients.
Keywords: Disease-modifying therapy; Multiple sclerosis; SARS-CoV-2; Third; Vaccination.
Copyright © 2022. Published by Elsevier B.V.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests:
Figures

References
-
- Achiron A., Mandel M., Dreyer-Alster S., Harari G., Magalashvili D., Sonis P., Dolev M., Menascu S., Flechter S., Falb R., Gurevich M. Humoral immune response to COVID-19 mRNA vaccine in patients with multiple sclerosis treated with high-efficacy disease-modifying therapies. Ther. Adv. Neurol. Disord. 2021;14 - PMC - PubMed
-
- Atmar, R.L., Lyke, K.E., Deming, M.E., Jackson, L.A., Branche, A.R., El Sahly, H.M., Rostad, C.A., Martin, J.M., Johnston, C., Rupp, R.E., Mulligan, M.J., Brady, R.C., Frenck, R.W., Backer, M., Kottkamp, A.C., Babu, T.M., Rajakumar, K., Edupuganti, S., Dobryzynski, D., Posavad, C.M., Archer, J.I., Crandon, S., Nayak, S.U., Szydlo, D., Zemanek, J., Islas, C.P.D., Brown, E.R., Suthar, M.S., McElrath, M.J., McDermott, A.B., O'Connell, S.E., Montefiori, D.C., Eaton, A., Neuzil, K.M., Stephens, D.S., Roberts, P.C., Beigel, J.H. Group, D.S., 2021. Heterologous SARS-CoV-2 Booster Vaccinations - Preliminary Report. medRxiv.
-
- Bonelli M., Mrak D., Tobudic S., Sieghart D., Koblischke M., Mandl P., Kornek B., Simader E., Radner H., Perkmann T., Haslacher H., Mayer M., Hofer P., Redlich K., Husar-Memmer E., Fritsch-Stork R., Thalhammer R., Stiasny K., Winkler S., Smolen J.S., Aberle J.H., Zeitlinger M., Heinz L.X., Aletaha D. Additional heterologous versus homologous booster vaccination in immunosuppressed patients without SARS-CoV-2 antibody seroconversion after primary mRNA vaccination: a randomised controlled trial. Ann. Rheum. Dis. 2022;81:687–694. - PubMed
-
- Bsteh G., Gradl C., Assar H., Heschl B., Hegen H., Di Pauli F., Leutmezer F., Traxler G., Zulehner G., Rommer P., Hiller M.S., Krajnc N., Wipfler P., Guger M., Enzinger C., Berger T. ÖGN Jahrestagung; Graz: 2022. COVID-19 in Multiple sclerosis: Update from the Nation-Wide Austrian registry.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

