Molecular approaches for spinal cord injury treatment
- PMID: 35799504
- PMCID: PMC9241396
- DOI: 10.4103/1673-5374.344830
Molecular approaches for spinal cord injury treatment
Abstract
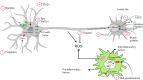
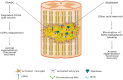
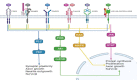
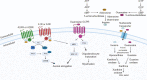
Injuries to the spinal cord result in permanent disabilities that limit daily life activities. The main reasons for these poor outcomes are the limited regenerative capacity of central neurons and the inhibitory milieu that is established upon traumatic injuries. Despite decades of research, there is still no efficient treatment for spinal cord injury. Many strategies are tested in preclinical studies that focus on ameliorating the functional outcomes after spinal cord injury. Among these, molecular compounds are currently being used for neurological recovery, with promising results. These molecules target the axon collapsed growth cone, the inhibitory microenvironment, the survival of neurons and glial cells, and the re-establishment of lost connections. In this review we focused on molecules that are being used, either in preclinical or clinical studies, to treat spinal cord injuries, such as drugs, growth and neurotrophic factors, enzymes, and purines. The mechanisms of action of these molecules are discussed, considering traumatic spinal cord injury in rodents and humans.
Keywords: axonal regeneration; drugs; enzymes; growth factors; molecular therapy; neurotrophic factors; purines; spinal cord injury.
Conflict of interest statement
None
Figures
References
-
- Anderson KD. Targeting recovery:priorities of the spinal cord-injured population. J Neurotrauma. 2004;21:1371–1383. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

