Expression, Purification, and in vitro Enzyme Activity Assay of a Recombinant Aldehyde Dehydrogenase from Thermus thermophilus, Using an Escherichia coli Host
- PMID: 35800460
- PMCID: PMC9090581
- DOI: 10.21769/BioProtoc.4401
Expression, Purification, and in vitro Enzyme Activity Assay of a Recombinant Aldehyde Dehydrogenase from Thermus thermophilus, Using an Escherichia coli Host
Abstract
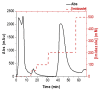
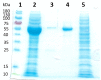
Based on previous in-depth characterisation, aldehyde dehydrogenases (ALDH) are a diverse superfamily of enzymes, in terms of both structure and function, present in all kingdoms of life. They catalyse the oxidation of an aldehyde to carboxylic acid using the cofactor nicotinamide adenine dinucleotide (phosphate) (NAD(P)+), and are often not substrate-specific, but rather have a broad range of associated biological functions, including detoxification and biosynthesis. We studied the structure of ALDHTt from Thermus thermophilus, as well as performed its biochemical characterisation. This allowed for insight into its potential substrates and biological roles. In this protocol, we describe ALDHTt heterologous expression in E. coli, purification, and activity assay (based on Shortall et al., 2021 ). ALDHTt was first copurified as a contaminant during caa3-type cytochrome oxidase isolation from T. thermophilus. This recombinant production system was employed for structural and biochemical analysis of wild-type and mutants, and proved efficient, yielding approximately 15-20 mg/L ALDHTt. For purification of the thermophilic his-tagged ALDHTt, heat treatment, immobilized metal affinity chromatography (IMAC), and gel filtration chromatography were used. The enzyme activity assay was performed via UV-Vis spectrophotometry, monitoring the production of reduced nicotinamide adenine dinucleotide (NADH). Graphical abstract: Flow chart outlining the steps in ALDHTt expression and purification, highlighting the approximate time required for each step.
Keywords: Aldehyde dehydrogenase; Auto-induction media; Cell culture; Enzymatic activity; Gel filtration chromatography; Heat treatment purification; Nickel affinity chromatography; UV-vis spectrophotometry.
Copyright © 2022 The Authors; exclusive licensee Bio-protocol LLC.
Conflict of interest statement
Competing interestsThere are no conflicts of interest or competing interests.
Figures
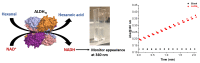

References
-
- Soulimane T.(2010). Thermus thermophilus encodes an archaeal-like fructose-1,6-bisphosphatase: purification of native and recombinant protein for structural studies. Protein Expr Purif 74(2): 175-180. - PubMed
LinkOut - more resources
Full Text Sources

