Two Sides of the Same Coin: Protein Kinase C γ in Cancer and Neurodegeneration
- PMID: 35800893
- PMCID: PMC9253466
- DOI: 10.3389/fcell.2022.929510
Two Sides of the Same Coin: Protein Kinase C γ in Cancer and Neurodegeneration
Abstract
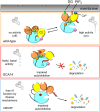
Protein kinase C (PKC) isozymes transduce myriad signals within the cell in response to the generation of second messengers from membrane phospholipids. The conventional isozyme PKCγ reversibly binds Ca2+ and diacylglycerol, which leads to an open, active conformation. PKCγ expression is typically restricted to neurons, but evidence for its expression in certain cancers has emerged. PKC isozymes have been labeled as oncogenes since the discovery that they bind tumor-promoting phorbol esters, however, studies of cancer-associated PKC mutations and clinical trial data showing that PKC inhibitors have worsened patient survival have reframed PKC as a tumor suppressor. Aberrant expression of PKCγ in certain cancers suggests a role outside the brain, although whether PKCγ also acts as a tumor suppressor remains to be established. On the other hand, PKCγ variants associated with spinocerebellar ataxia type 14 (SCA14), a neurodegenerative disorder characterized by Purkinje cell degeneration, enhance basal activity while preventing phorbol ester-mediated degradation. Although the basis for SCA14 Purkinje cell degeneration remains unknown, studies have revealed how altered PKCγ activity rewires cerebellar signaling to drive SCA14. Importantly, enhanced basal activity of SCA14-associated mutants inversely correlates with age of onset, supporting that enhanced PKCγ activity drives SCA14. Thus, PKCγ activity should likely be inhibited in SCA14, whereas restoring PKC activity should be the goal in cancer therapies. This review describes how PKCγ activity can be lost or gained in disease and the overarching need for a PKC structure as a powerful tool to predict the effect of PKCγ mutations in disease.
Keywords: autoinhibition; cancer; neurodegeneration; protein kinase C; spinocerebellar ataxia.
Copyright © 2022 Pilo and Newton.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

