A Proteinaceous Alpha-Amylase Inhibitor from Moringa Oleifera Leaf Extract: Purification, Characterization, and Insecticide Effects against C. maculates Insect Larvae
- PMID: 35807466
- PMCID: PMC9268253
- DOI: 10.3390/molecules27134222
A Proteinaceous Alpha-Amylase Inhibitor from Moringa Oleifera Leaf Extract: Purification, Characterization, and Insecticide Effects against C. maculates Insect Larvae
Abstract
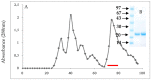
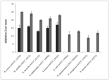
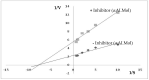
The main objective of the current study was the extraction, purification, and enzymatic characterization of a potent proteinaceous amylase inhibitor from Moringa oleifera. The antimicrobial potential and insecticide effects against C. maculates insect larvae were also studied. The α-amylase inhibitor was extracted in methanol (with an inhibitory activity of 65.6% ± 4.93). Afterwards, the inhibitor αAI.Mol was purified after a heat treatment at 70 °C for 15 min followed by one chromatographic step of Sephadex G-50. An apparent molecular weight of 25 kDa was analyzed, and the N-terminal sequence showed the highest identity level (89%) with the monomeric α-amylase inhibitor from Triticum dicoccoides. αAI.Mol was found to tolerate pH values ranging from 5.0 to 11.0 and showed maximal activity at pH 9.0. Thermal stability was remarkably important, since the inhibitory activity was maintained at 55% after 1 h of incubation at 70 °C and at 53% after an incubation of 45 min at 80 °C. The potency of the current purified inhibitor against amylases from different origins indicates that αAI.Mol seems to possess the highest affinity toward human salivary α-amylase (90% inhibitory activity), followed by the α-amylase of insects Callosobruchus maculatus and Tribolium confusum (71% and 61%, respectively). The kinetic parameters were also calculated, and the Kmax and Vmax of the digestive amylase were estimated at 185 (mmol/min/mg) and 0.13 mM, respectively. The inhibitor possesses a strong bactericidal effect against Gram+ and Gram- strains, and the MIC values were >1 against B. cereus but >6 against E. coli. Interestingly, the rates of survival and pupation of C. maculates insect larvae were remarkably affected by the purified αAI.Mol from Moringa oleifera.
Keywords: insecticide effects; kinetic parameters; plant defense; α-amylase inhibitor.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


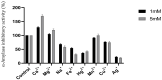

References
-
- Gallienne E. Ph.D. Thesis. Universite Blaise Pascal; Aubière, France: 2000. Synthèse et évaluation d’inhibiteurs potentiels de glycosidases, analogues du salacinol.
-
- Konarev A.V. Interaction of insect digestive enzymes with plant protein inhibitors and host-parasite co-evolution. Euphytica. 1996;92:89–94. doi: 10.1007/BF00022833. - DOI
-
- Chrispeels M.J., Grossid S.M.F., Higgins T.J.V. Genetic engineering with a-amylase inhibitors seeds resistant to bruchids. Seed Sci. Res. 1998;8:257–263. doi: 10.1017/S0960258500004153. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

