Volumetric analysis of the piriform cortex in temporal lobe epilepsy
- PMID: 35810570
- PMCID: PMC10510027
- DOI: 10.1016/j.eplepsyres.2022.106971
Volumetric analysis of the piriform cortex in temporal lobe epilepsy
Abstract
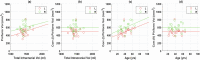
The piriform cortex, at the confluence of the temporal and frontal lobes, generates seizures in response to chemical convulsants and electrical stimulation. Resection of more than 50% of the piriform cortex in anterior temporal lobe resection for refractory temporal lobe epilepsy (TLE) was associated with a 16-fold higher chance of seizure freedom. The objectives of the current study were to implement a robust protocol to measure piriform cortex volumes and to quantify the correlation of these volumes with clinical characteristics of TLE. Sixty individuals with unilateral TLE (33 left) and 20 healthy controls had volumetric analysis of left and right piriform cortex and hippocampi. A protocol for segmenting and measuring the volumes of the piriform cortices was implemented, with good inter-rater and test-retest reliability. The right piriform cortex volume was consistently larger than the left piriform cortex in both healthy controls and patients with TLE. In controls, the mean volume of the right piriform cortex was 17.7% larger than the left, and the right piriform cortex extended a mean of 6 mm (Range: -4 to 12) more anteriorly than the left. This asymmetry was also seen in left and right TLE. In TLE patients overall, the piriform cortices were not significantly smaller than in controls. Hippocampal sclerosis was associated with decreased ipsilateral and contralateral piriform cortex volumes. The piriform cortex volumes, both ipsilateral and contralateral to the epileptic temporal lobe, were smaller with a longer duration of epilepsy. There was no significant association between piriform cortex volumes and the frequency of focal seizures with impaired awareness or the number of anti-seizure medications taken. Implementation of robust segmentation will enable consistent neurosurgical resection in anterior temporal lobe surgery for refractory TLE..
Keywords: Hippocampus; Piriform cortex; Temporal lobe epilepsy.
Copyright © 2022. Published by Elsevier B.V.
Conflict of interest statement
None.
Figures




Similar articles
-
Association of Piriform Cortex Resection With Surgical Outcomes in Patients With Temporal Lobe Epilepsy.JAMA Neurol. 2019 Jun 1;76(6):690-700. doi: 10.1001/jamaneurol.2019.0204. JAMA Neurol. 2019. PMID: 30855662 Free PMC article.
-
Cerebrocerebellar structural covariance in temporal lobe epilepsy with hippocampal sclerosis.Epilepsy Behav. 2020 Oct;111:107180. doi: 10.1016/j.yebeh.2020.107180. Epub 2020 Jun 26. Epilepsy Behav. 2020. PMID: 32599430
-
Resection of the piriform cortex for temporal lobe epilepsy: a Novel approach on imaging segmentation and surgical application.Br J Neurosurg. 2024 Jun;38(3):716-721. doi: 10.1080/02688697.2021.1966385. Epub 2021 Aug 18. Br J Neurosurg. 2024. PMID: 34406102
-
Feline temporal lobe epilepsy: seven cases of hippocampal and piriform lobe necrosis in England and literature review.J Feline Med Surg. 2022 Jun;24(6):596-608. doi: 10.1177/1098612X211035049. Epub 2021 Aug 6. J Feline Med Surg. 2022. PMID: 34355984 Free PMC article. Review.
-
The potential therapeutic role of berberine in treating epilepsy focusing on temporal lobe epilepsy: State of art and ongoing perspective.Brain Res Bull. 2025 Feb;221:111189. doi: 10.1016/j.brainresbull.2025.111189. Epub 2025 Jan 4. Brain Res Bull. 2025. PMID: 39761924 Review.
Cited by
-
The role of the piriform cortex in temporal lobe epilepsy: A current literature review.Front Neurol. 2022 Nov 21;13:1042887. doi: 10.3389/fneur.2022.1042887. eCollection 2022. Front Neurol. 2022. PMID: 36479052 Free PMC article. Review.
-
The Little-Known Ribbon-Shaped Piriform Cortex: A Key Node in Temporal Lobe Epilepsy-Anatomical Insights and Its Potential for Surgical Treatment.Diagnostics (Basel). 2024 Dec 17;14(24):2838. doi: 10.3390/diagnostics14242838. Diagnostics (Basel). 2024. PMID: 39767200 Free PMC article. Review.
-
Relationship of left piriform cortex network centrality with temporal lobe epilepsy duration and drug resistance.Eur J Neurol. 2025 Feb;32(2):e70018. doi: 10.1111/ene.70018. Eur J Neurol. 2025. PMID: 39949073 Free PMC article.
-
Piriform cortex is an ictogenic trigger zone in the primate brain.Epilepsia. 2025 Feb;66(2):569-582. doi: 10.1111/epi.18201. Epub 2024 Dec 5. Epilepsia. 2025. PMID: 39636294
-
Extent of piriform cortex resection in children with temporal lobe epilepsy.Ann Clin Transl Neurol. 2023 Sep;10(9):1613-1622. doi: 10.1002/acn3.51852. Epub 2023 Jul 20. Ann Clin Transl Neurol. 2023. PMID: 37475156 Free PMC article.
References
-
- Alhusaini S., Doherty C.P., Scanlon C., Ronan L., Maguire S., Borgulya G., et al. A cross-sectional MRI study of brain regional atrophy and clinical characteristics of temporal lobe epilepsy with hippocampal sclerosis. Epilepsy Res. 2012;99:156–166. - PubMed
-
- Bernasconi N., Bernasconi A., Caramanos Z., Antel S.B., Andermann F., Arnold D.L. Mesial temporal damage in temporal lobe epilepsy: A volumetric MRI study of the hippocampus, amygdala and parahippocampal region. Brain. 2003;126:462–469. - PubMed
-
- Bland J.M., Altman D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;1:307–310. - PubMed

