Dihydroartemisinin regulates immune cell heterogeneity by triggering a cascade reaction of CDK and MAPK phosphorylation
- PMID: 35811310
- PMCID: PMC9271464
- DOI: 10.1038/s41392-022-01028-5
Dihydroartemisinin regulates immune cell heterogeneity by triggering a cascade reaction of CDK and MAPK phosphorylation
Abstract
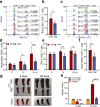
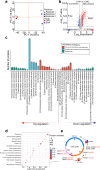
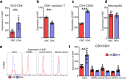
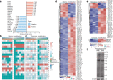
Artemisinin (ART) and dihydroartemisinin (DHA), apart from their profound anti-malaria effect, can also beneficially modulate the host immune system; however, the underlying molecular mechanisms remain unclear. Here, we report that DHA selectively induced T-cell activation, with an increased proportion of Ki67+CD4+ T cells, CD25+CD4+ T cells, interferon (IFN)-γ-producing CD8+ T cells, Brdu+ CD8+ T cells and neutrophils, which was found to enhance cellular immunity to experimental malaria and overcome immunosuppression in mice. We further revealed that DHA upregulated the expression of cell proliferation-associated proteins by promoting the phosphorylation of mitogen-activated protein kinase (MAPK), cyclin-dependent kinases (CDKs), and activator protein 1 in the spleen. This study is the first to provide robust evidence that DHA selectively induced the expansion of subsets of splenic T cells through phosphorylated CDKs and MAPK to enhance cellular immune responses under non-pathological or pathological conditions. The data significantly deepened our knowledge in the mechanism underlying DHA-mediated immunomodulation.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Dihydroartemisinin beneficially regulates splenic immune cell heterogeneity through the SOD3-JNK-AP-1 axis.Sci China Life Sci. 2022 Aug;65(8):1636-1654. doi: 10.1007/s11427-021-2061-7. Epub 2022 Feb 23. Sci China Life Sci. 2022. PMID: 35226255
-
Dihydroartemisinin regulates the immune system by promotion of CD8+ T lymphocytes and suppression of B cell responses.Sci China Life Sci. 2020 May;63(5):737-749. doi: 10.1007/s11427-019-9550-4. Epub 2019 Jul 8. Sci China Life Sci. 2020. PMID: 31290095
-
Dihydroartemisinin inhibits melanoma by regulating CTL/Treg anti-tumor immunity and STAT3-mediated apoptosis via IL-10 dependent manner.J Dermatol Sci. 2020 Sep;99(3):193-202. doi: 10.1016/j.jdermsci.2020.08.001. Epub 2020 Aug 9. J Dermatol Sci. 2020. PMID: 32859456
-
JNK and p38 MAP kinases in CD4+ and CD8+ T cells.Immunol Rev. 2003 Apr;192:131-42. doi: 10.1034/j.1600-065x.2003.00019.x. Immunol Rev. 2003. PMID: 12670401 Review.
-
Dihydroartemisinin: A Potential Natural Anticancer Drug.Int J Biol Sci. 2021 Jan 16;17(2):603-622. doi: 10.7150/ijbs.50364. eCollection 2021. Int J Biol Sci. 2021. PMID: 33613116 Free PMC article. Review.
Cited by
-
Ly6G+ Neutrophils and Interleukin-17 Are Essential in Protection against Rodent Malaria Caused by Plasmodium berghei ANKA.Research (Wash D C). 2024 Dec 19;7:0559. doi: 10.34133/research.0559. eCollection 2024. Research (Wash D C). 2024. PMID: 39703777 Free PMC article.
-
A sonoelectric niche for noninvasive intervertebral disc regeneration via targeted cell cycle modulation.Sci Adv. 2025 Aug 8;11(32):eadu6860. doi: 10.1126/sciadv.adu6860. Epub 2025 Aug 8. Sci Adv. 2025. PMID: 40779637 Free PMC article.
-
Medicinal plant-derived mtDNA via nanovesicles induces the cGAS-STING pathway to remold tumor-associated macrophages for tumor regression.J Nanobiotechnology. 2023 Mar 6;21(1):78. doi: 10.1186/s12951-023-01835-0. J Nanobiotechnology. 2023. PMID: 36879291 Free PMC article.
-
Prevotella copri exhausts intrinsic indole-3-pyruvic acid in the host to promote breast cancer progression: inactivation of AMPK via UHRF1-mediated negative regulation.Gut Microbes. 2024 Jan-Dec;16(1):2347757. doi: 10.1080/19490976.2024.2347757. Epub 2024 May 21. Gut Microbes. 2024. PMID: 38773738 Free PMC article.
-
Artemisinin derivatives differently affect cell death of lung cancer subtypes by regulating GPX4 in patient-derived tissue cultures.Cell Death Discov. 2025 May 28;11(1):256. doi: 10.1038/s41420-025-02537-2. Cell Death Discov. 2025. PMID: 40436830 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

