A Temporary Immersion System to Improve Cannabis sativa Micropropagation
- PMID: 35812929
- PMCID: PMC9262383
- DOI: 10.3389/fpls.2022.895971
A Temporary Immersion System to Improve Cannabis sativa Micropropagation
Abstract
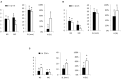
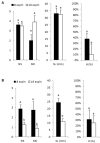
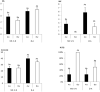
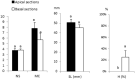
The aim of this study was to propagate axillary shoots of Cannabis sativa L. using liquid medium in temporary immersion bioreactors. The effect of immersion frequency (3 or 6 immersions per day), explant type (apical or basal sections), explant number (8, 10, and 16 explants), mineral medium (Murashige and Skoog half-strength nitrates, β-A and β-H, all supplemented with 2-μM metatopoline), sucrose supplementation (2, 0.5, and 0% sucrose), culture duration (4 and 6 weeks), and bioreactor type (RITA® and Plantform™) were investigated. As a result, we propose a protocol for the proliferation of cannabis apical segments in RITA® or Plantform™ bioreactors. The explants (8 per RITA® and 24 per Plantform™) are immersed for 1 min, 3 times per day in β-A medium supplemented with 2-μM metatopoline and 0.5% of sucrose and subcultured every 4 weeks. This is the first study using temporary immersion systems in C. sativa production, and our results provide new opportunities for the mass propagation of this species.
Keywords: Plantform™; RITA; bioreactors; liquid medium; mass propagation; sucrose.
Copyright © 2022 Rico, Garrido, Sánchez, Ferreiro-Vera, Codesido and Vidal.
Conflict of interest statement
JG, CF-V, and VC are employed by Phytoplant Research SLU. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Aitken-Christie J. (1991). Automation, in Micropropagation, eds Debergh P. C., Zimmerman R. H. (Dordrecht: Springer; ), 363–388. 10.1007/978-94-009-2075-0_23 - DOI
-
- Akdemir H., Süzerer V., Onay A., Tilkat E., Ersali Y., Çiftçi Y. O. (2014). Micropropagation of the pistachio and its rootstocks by temporary immersion system. Plant Cell. Tissue Organ Cult. 117, 65–76. 10.1007/s11240-013-0421-0 - DOI
-
- Akin M., Eyduran E., Reed B. M. (2017). Use of RSM and CHAID data mining algorithm for predicting mineral nutrition of hazelnut. Plant Cell Tissue Organ Cult. 128, 303–316. 10.1007/s11240-016-1110-6 - DOI
-
- Albarrán J., Bertrand B., Lartaud M., Etienne H. (2005). Cycle characteristics in a temporary immersion bioreactor affect regeneration, morphology, water and mineral status of coffee (Coffea arabica) somatic embryos. Plant Cell. Tissue Organ Cult. 81, 27–36. 10.1007/s11240-004-2618-8 - DOI
LinkOut - more resources
Full Text Sources
Research Materials

