Marsupials and Multi-Omics: Establishing New Comparative Models of Neural Crest Patterning and Craniofacial Development
- PMID: 35813210
- PMCID: PMC9260703
- DOI: 10.3389/fcell.2022.941168
Marsupials and Multi-Omics: Establishing New Comparative Models of Neural Crest Patterning and Craniofacial Development
Abstract
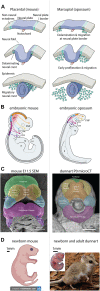
Studies across vertebrates have revealed significant insights into the processes that drive craniofacial morphogenesis, yet we still know little about how distinct facial morphologies are patterned during development. Studies largely point to evolution in GRNs of cranial progenitor cell types such as neural crest cells, as the major driver underlying adaptive cranial shapes. However, this hypothesis requires further validation, particularly within suitable models amenable to manipulation. By utilizing comparative models between related species, we can begin to disentangle complex developmental systems and identify the origin of species-specific patterning. Mammals present excellent evolutionary examples to scrutinize how these differences arise, as sister clades of eutherians and marsupials possess suitable divergence times, conserved cranial anatomies, modular evolutionary patterns, and distinct developmental heterochrony in their NCC behaviours and craniofacial patterning. In this review, I lend perspectives into the current state of mammalian craniofacial biology and discuss the importance of establishing a new marsupial model, the fat-tailed dunnart, for comparative research. Through detailed comparisons with the mouse, we can begin to decipher mammalian conserved, and species-specific processes and their contribution to craniofacial patterning and shape disparity. Recent advances in single-cell multi-omics allow high-resolution investigations into the cellular and molecular basis of key developmental processes. As such, I discuss how comparative evolutionary application of these tools can provide detailed insights into complex cellular behaviours and expression dynamics underlying adaptive craniofacial evolution. Though in its infancy, the field of "comparative evo-devo-omics" presents unparalleled opportunities to precisely uncover how phenotypic differences arise during development.
Keywords: GRN; NCC; constraint; evolution; heterochrony; mammal; skull.
Copyright © 2022 Newton.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Skull Development, Ossification Pattern, and Adult Shape in the Emerging Lizard Model Organism Pogona vitticeps: A Comparative Analysis With Other Squamates.Front Physiol. 2018 Mar 28;9:278. doi: 10.3389/fphys.2018.00278. eCollection 2018. Front Physiol. 2018. PMID: 29643813 Free PMC article.
-
Molecular mechanisms of cranial neural crest cell migration and patterning in craniofacial development.Development. 2010 Aug;137(16):2605-21. doi: 10.1242/dev.040048. Development. 2010. PMID: 20663816 Review.
-
Early differentiation and migration of cranial neural crest in the opossum, Monodelphis domestica.Evol Dev. 2003 Mar-Apr;5(2):121-35. doi: 10.1046/j.1525-142x.2003.03019.x. Evol Dev. 2003. PMID: 12622729
-
Recent advances in craniofacial morphogenesis.Dev Dyn. 2006 Sep;235(9):2353-75. doi: 10.1002/dvdy.20833. Dev Dyn. 2006. PMID: 16680722 Review.
-
Craniofacial development in marsupial mammals: developmental origins of evolutionary change.Dev Dyn. 2006 May;235(5):1181-93. doi: 10.1002/dvdy.20676. Dev Dyn. 2006. PMID: 16408286 Review.
Cited by
-
Embryology of the fat-tailed dunnart (Sminthopsis crassicaudata): A marsupial model for comparative mammalian developmental and evolutionary biology.Dev Dyn. 2025 Feb;254(2):142-157. doi: 10.1002/dvdy.711. Epub 2024 May 9. Dev Dyn. 2025. PMID: 38721717 Free PMC article.
References
-
- Brugmann S. A., Powder K. E., Young N. M., Goodnough L. H., Hahn S. M., James A. W., et al. (2010). Comparative Gene Expression Analysis of Avian Embryonic Facial Structures Reveals New Candidates for Human Craniofacial Disorders. Hum. Mol. Genet. 19, 920–930. 10.1093/hmg/ddp559 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

