Opposing Roles of Wild-type and Mutant p53 in the Process of Epithelial to Mesenchymal Transition
- PMID: 35813818
- PMCID: PMC9261265
- DOI: 10.3389/fmolb.2022.928399
Opposing Roles of Wild-type and Mutant p53 in the Process of Epithelial to Mesenchymal Transition
Abstract
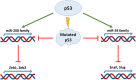
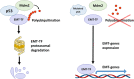
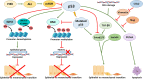
The central role of an aberrantly activated EMT program in defining the critical features of aggressive carcinomas is well documented and includes cell plasticity, metastatic dissemination, drug resistance, and cancer stem cell-like phenotypes. The p53 tumor suppressor is critical for leashing off all the features mentioned above. On the molecular level, the suppression of these effects is exerted by p53 via regulation of its target genes, whose products are involved in cell cycle, apoptosis, autophagy, DNA repair, and interactions with immune cells. Importantly, a set of specific mutations in the TP53 gene (named Gain-of-Function mutations) converts this tumor suppressor into an oncogene. In this review, we attempted to contrast different regulatory roles of wild-type and mutant p53 in the multi-faceted process of EMT.
Keywords: EMT; epigenetic regulation; epithelial to mesenchymal transition; microRNA; mutant p53; p53; wild-type p53.
Copyright © 2022 Semenov, Daks, Fedorova, Shuvalov and Barlev.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Inhibition of glucosylceramide synthase eliminates the oncogenic function of p53 R273H mutant in the epithelial-mesenchymal transition and induced pluripotency of colon cancer cells.Oncotarget. 2016 Sep 13;7(37):60575-60592. doi: 10.18632/oncotarget.11169. Oncotarget. 2016. PMID: 27517620 Free PMC article.
-
Mutant p53(R175H) upregulates Twist1 expression and promotes epithelial-mesenchymal transition in immortalized prostate cells.Cell Death Differ. 2011 Feb;18(2):271-81. doi: 10.1038/cdd.2010.94. Epub 2010 Aug 6. Cell Death Differ. 2011. PMID: 20689556 Free PMC article.
-
Antagonistic Effects of p53 and HIF1A on microRNA-34a Regulation of PPP1R11 and STAT3 and Hypoxia-induced Epithelial to Mesenchymal Transition in Colorectal Cancer Cells.Gastroenterology. 2017 Aug;153(2):505-520. doi: 10.1053/j.gastro.2017.04.017. Epub 2017 Apr 20. Gastroenterology. 2017. PMID: 28435028
-
Crosslink between p53 and metastasis: focus on epithelial-mesenchymal transition, cancer stem cell, angiogenesis, autophagy, and anoikis.Mol Biol Rep. 2021 Nov;48(11):7545-7557. doi: 10.1007/s11033-021-06706-1. Epub 2021 Sep 14. Mol Biol Rep. 2021. PMID: 34519942 Review.
-
Cancer Stemness: p53 at the Wheel.Front Oncol. 2021 Jan 11;10:604124. doi: 10.3389/fonc.2020.604124. eCollection 2020. Front Oncol. 2021. PMID: 33505918 Free PMC article. Review.
Cited by
-
ZEB1 hypermethylation is associated with better prognosis in patients with colon cancer.Clin Epigenetics. 2023 Dec 13;15(1):193. doi: 10.1186/s13148-023-01605-7. Clin Epigenetics. 2023. PMID: 38093305 Free PMC article.
-
PTC596-Induced BMI-1 Inhibition Fights Neuroblastoma Multidrug Resistance by Inducing Ferroptosis.Antioxidants (Basel). 2023 Dec 19;13(1):3. doi: 10.3390/antiox13010003. Antioxidants (Basel). 2023. PMID: 38275623 Free PMC article.
-
Expression of E-Cadherin and N-Cadherin in the Endocervix as a Predictive Factor in Patients with Endometrial Cancer.Int J Mol Sci. 2024 Mar 21;25(6):3547. doi: 10.3390/ijms25063547. Int J Mol Sci. 2024. PMID: 38542521 Free PMC article.
-
Cellular and Molecular Mechanisms of the Tumor Stroma in Colorectal Cancer: Insights into Disease Progression and Therapeutic Targets.Biomedicines. 2023 Aug 23;11(9):2361. doi: 10.3390/biomedicines11092361. Biomedicines. 2023. PMID: 37760801 Free PMC article. Review.
-
Alpha lipoic acid diminishes migration and invasion in hepatocellular carcinoma cells through an AMPK-p53 axis.Sci Rep. 2024 Sep 11;14(1):21275. doi: 10.1038/s41598-024-72309-y. Sci Rep. 2024. PMID: 39261583 Free PMC article.
References
-
- Alam S. K., Yadav V. K., Bajaj S., Datta A., Dutta S. K., Bhattacharyya M., et al. (2016). DNA Damage-Induced Ephrin-B2 Reverse Signaling Promotes Chemoresistance and Drives EMT in Colorectal Carcinoma Harboring Mutant P53. Cell Death Differ. 23 (4), 707–722. 10.1038/cdd.2015.133 - DOI - PMC - PubMed
-
- Alvarado-Ortiz E., de la Cruz-López K. G., Becerril-Rico J., Sarabia-Sánchez M. A., Ortiz-Sánchez E., García-Carrancá A. (2021). Mutant P53 Gain-Of-Function: Role in Cancer Development, Progression, and Therapeutic Approaches. Front. Cell Dev. Biol. 8, 1868. 10.3389/fcell.2020.607670 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

