Bladder Acellular Matrix Prepared by a Self-Designed Perfusion System and Adipose-Derived Stem Cells to Promote Bladder Tissue Regeneration
- PMID: 35814010
- PMCID: PMC9257038
- DOI: 10.3389/fbioe.2022.794603
Bladder Acellular Matrix Prepared by a Self-Designed Perfusion System and Adipose-Derived Stem Cells to Promote Bladder Tissue Regeneration
Abstract
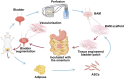
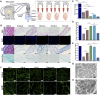
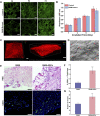
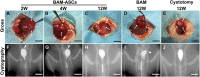
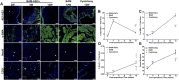
The bladder patch constructed with the bladder acellular matrix (BAM) and adipose-derived stem cells (ASCs) was incubated with the omentum for bladder reconstruction in a rat model of bladder augmentation cystoplasty. A self-designed perfusion system and five different decellularization protocols were used to prepare the BAM. Finally, an optimal protocol (group C) was screened out by comparing the cell nucleus residue, collagen structure preservation and biologically active components retention of the prepared BAM. ASCs-seeded (BAM-ASCs group) and unseeded BAM (BAM group) were incubated with the omentum for 7 days to promote neovascularization and then perform bladder reconstruction. Hematoxylin and eosin and Masson's trichrome staining indicated that the bladder patches in the BAM-ASCs group could better regenerate the bladder wall structure compared to the BAM group. Moreover, immunofluorescence analyses demonstrated that the ASCs could promote the regeneration of smooth muscle, neurons and blood vessels, and the physiological function (maximal bladder capacity, max pressure prior to voiding and bladder compliance) restoration in the BAM-ASCs group. The results demonstrated that the self-designed perfusion system could quickly and efficiently prepare the whole bladder scaffold and confirmed that the prepared BAM could be used as the scaffold material for functional bladder tissue engineering applications.
Keywords: adipose-derived stem cells; bladder acellular matrix; perfusion system; tissue engineering; vascularization.
Copyright © 2022 Xiao, Wang, Zhao, Ling, An, Fu, Fu, Zhou and Zhang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

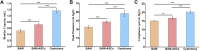
Similar articles
-
Bi-layer silk fibroin skeleton and bladder acellular matrix hydrogel encapsulating adipose-derived stem cells for bladder reconstruction.Biomater Sci. 2021 Sep 14;9(18):6169-6182. doi: 10.1039/d1bm00761k. Biomater Sci. 2021. PMID: 34346416
-
Bladder Acellular Matrix Grafts Seeded with Adipose-Derived Stem Cells and Incubated Intraperitoneally Promote the Regeneration of Bladder Smooth Muscle and Nerve in a Rat Model of Bladder Augmentation.Stem Cells Dev. 2016 Mar 1;25(5):405-14. doi: 10.1089/scd.2015.0246. Epub 2016 Feb 10. Stem Cells Dev. 2016. PMID: 26863067
-
The morphological regeneration and functional restoration of bladder defects by a novel scaffold and adipose-derived stem cells in a rat augmentation model.Stem Cell Res Ther. 2017 Jun 24;8(1):149. doi: 10.1186/s13287-017-0597-z. Stem Cell Res Ther. 2017. PMID: 28646909 Free PMC article.
-
Application of bladder acellular matrix in urinary bladder regeneration: the state of the art and future directions.Biomed Res Int. 2015;2015:613439. doi: 10.1155/2015/613439. Epub 2015 Feb 22. Biomed Res Int. 2015. PMID: 25793199 Free PMC article. Review.
-
Bladder acellular matrix and its application in bladder augmentation.Tissue Eng Part B Rev. 2014 Apr;20(2):163-72. doi: 10.1089/ten.TEB.2013.0103. Epub 2013 Sep 21. Tissue Eng Part B Rev. 2014. PMID: 23895225 Review.
Cited by
-
A bilayer bioengineered patch with sequential dual-growth factor release to promote vascularization in bladder reconstruction.Regen Biomater. 2024 Jul 25;11:rbae083. doi: 10.1093/rb/rbae083. eCollection 2024. Regen Biomater. 2024. PMID: 39077683 Free PMC article.
-
Bioactive scaffolds for tissue engineering: A review of decellularized extracellular matrix applications and innovations.Exploration (Beijing). 2024 Apr 11;5(1):20230078. doi: 10.1002/EXP.20230078. eCollection 2025 Feb. Exploration (Beijing). 2024. PMID: 40040827 Free PMC article. Review.
-
Use of adipose-derived stem cells on decellularized bladder scaffolds for functional bladder mucosa regeneration.Stem Cells Transl Med. 2025 Jun 25;14(7):szaf033. doi: 10.1093/stcltm/szaf033. Stem Cells Transl Med. 2025. PMID: 40627336 Free PMC article.
-
The BAM-GelMA-ADSCs bilayer patch promotes tissue regeneration and functional recovery after large-area bladder defects in beagles.Bioeng Transl Med. 2025 Jan 15;10(3):e10745. doi: 10.1002/btm2.10745. eCollection 2025 May. Bioeng Transl Med. 2025. PMID: 40385542 Free PMC article.
-
Perfusion preparation of the rat bladder decellularized scaffold.Regen Ther. 2023 Sep 26;24:499-506. doi: 10.1016/j.reth.2023.09.005. eCollection 2023 Dec. Regen Ther. 2023. PMID: 37779903 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

