Advanced in Vitro Safety Assessment of Herbal Medicines for the Treatment of Non-Psychotic Mental Disorders in Pregnancy
- PMID: 35814220
- PMCID: PMC9259859
- DOI: 10.3389/fphar.2022.882997
Advanced in Vitro Safety Assessment of Herbal Medicines for the Treatment of Non-Psychotic Mental Disorders in Pregnancy
Abstract
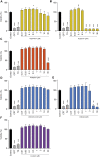
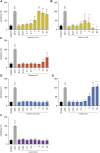
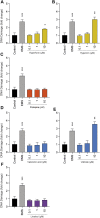
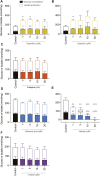
When confronted with non-psychotic mental disorders, pregnant women often refrain from using synthetic drugs and resort to herbal medicines such as St. John's wort, California poppy, valerian, lavender, and hops. Nevertheless, these herbal medicines have not yet been officially approved in pregnancy due to lack of safety data. Using a variety of in vitro methods (determination of cytotoxicity, apoptosis induction, genotoxicity, effects on metabolic properties, and inhibition/induction of differentiation) in a commonly used placental cell line (BeWo b30), we were previously able to show that extracts from these plants are likely to be safe at the usual clinical doses. In the present work, we wanted to extend our safety assessment of these herbal medicines by 1) looking for possible effects on gene expression and 2) using the same in vitro methods to characterize effects of selected phytochemicals that might conceivably lead to safety issues. Proteomics results were promising, as none of the five extracts significantly affected protein expression by up- or down-regulation. Protopine (contained in California poppy), valerenic acid (in valerian), and linalool (in lavender) were inconspicuous in all experiments and showed no adverse effects. Hyperforin and hypericin (two constituents of St. John's wort) and valtrate (typical for valerian) were the most obvious phytochemicals with respect to cytotoxic and apoptotic effects. A decrease in cell viability was evident with hypericin (≥1 µM) and valtrate (≥10 µM), whereas hyperforin (≥3 µM), hypericin (30 µM) and valtrate (≥10 µM) induced cell apoptosis. None of the tested phytochemicals resulted in genotoxic effects at concentrations of 0.1 and 1 µM and thus are not DNA damaging. No decrease in glucose consumption or lactate production was observed under the influence of the phytochemicals, except for valtrate (at all concentrations). No compound affected cell differentiation, except for hyperforin (≥1 µM), which had an inhibitory effect. This study suggests that extracts from St. John's wort, California poppy, valerian, lavender, and hops are likely to be safe during pregnancy. High plasma concentrations of some relevant compounds-hyperforin and hypericin from St. John's wort and valtrate from valerian-deserve special attention, however.
Keywords: Eschscholzia californica; Humulus lupulus; Hypericum perforatum; Lavandula angustifolia; Valeriana officinalis; mental health disorders; pregnancy; safety.
Copyright © 2022 Spiess, Winker, Dolder Behna, Gründemann and Simões-Wüst.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The handling editor MH declared a past co-authorship with the author CG.
Figures

Similar articles
-
Transplacental passage of hyperforin, hypericin, and valerenic acid.Front Pharmacol. 2023 Mar 31;14:1123194. doi: 10.3389/fphar.2023.1123194. eCollection 2023. Front Pharmacol. 2023. PMID: 37063288 Free PMC article.
-
Medicinal Plants for the Treatment of Mental Diseases in Pregnancy: An In Vitro Safety Assessment.Planta Med. 2022 Oct;88(12):1036-1046. doi: 10.1055/a-1628-8132. Epub 2021 Oct 8. Planta Med. 2022. PMID: 34624906 Free PMC article.
-
Immunological evaluation of herbal extracts commonly used for treatment of mental diseases during pregnancy.Sci Rep. 2023 Jun 14;13(1):9630. doi: 10.1038/s41598-023-35952-5. Sci Rep. 2023. PMID: 37316493 Free PMC article.
-
St John's wort (Hypericum perforatum L.): a review of its chemistry, pharmacology and clinical properties.J Pharm Pharmacol. 2001 May;53(5):583-600. doi: 10.1211/0022357011775910. J Pharm Pharmacol. 2001. PMID: 11370698 Review.
-
Understanding drug interactions with St John's wort (Hypericum perforatum L.): impact of hyperforin content.J Pharm Pharmacol. 2019 Jan;71(1):129-138. doi: 10.1111/jphp.12858. Epub 2018 Feb 7. J Pharm Pharmacol. 2019. PMID: 29411879 Review.
Cited by
-
Acute, chronic, and genotoxic studies on the protopine total alkaloids of the Macleaya cordata (willd.) R. Br. in rodents.Front Pharmacol. 2022 Sep 28;13:987800. doi: 10.3389/fphar.2022.987800. eCollection 2022. Front Pharmacol. 2022. PMID: 36249819 Free PMC article.
-
Transplacental passage of hyperforin, hypericin, and valerenic acid.Front Pharmacol. 2023 Mar 31;14:1123194. doi: 10.3389/fphar.2023.1123194. eCollection 2023. Front Pharmacol. 2023. PMID: 37063288 Free PMC article.
-
Safety and efficacy of a feed additive consisting of Macleaya cordata (Willd.) R. Br. extract and leaves (Sangrovit® extra) for all poultry species (excluding laying and breeding birds) (Phytobiotics Futterzusatzstoffe GmbH).EFSA J. 2023 Jun 9;21(6):e08052. doi: 10.2903/j.efsa.2023.8052. eCollection 2023 Jun. EFSA J. 2023. PMID: 37304353 Free PMC article.
-
Supplementary approaches to perinatal depression: a review of pathogenesis, herbal interventions, and dietary supplements.Front Psychol. 2025 May 21;16:1529339. doi: 10.3389/fpsyg.2025.1529339. eCollection 2025. Front Psychol. 2025. PMID: 40491948 Free PMC article. Review.
References
-
- Blaschek W. (2016). Wichtl - Teedrogen und Phytopharmaka. Stuttgart: Wissenschaftliche Verlagsgesellschaft mbH.
LinkOut - more resources
Full Text Sources

