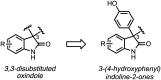
Evolution of 3-(4-hydroxyphenyl)indoline-2-one as a scaffold for potent and selective anticancer activity
- PMID: 35814932
- PMCID: PMC9215341
- DOI: 10.1039/d2md00110a
Evolution of 3-(4-hydroxyphenyl)indoline-2-one as a scaffold for potent and selective anticancer activity
Abstract
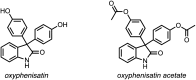
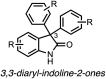
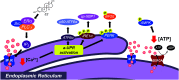
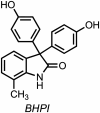
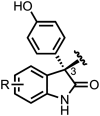
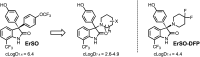
Development of targeted anticancer modalities has prompted a new era in cancer treatment that is notably different from the age of radical surgery and highly toxic chemotherapy. Behind each effective compound is a rich and complex history from first identification of chemical matter, detailed optimization, and mechanistic investigations, ultimately leading to exciting molecules for drug development. Herein we review the history and on-going journey of one such anticancer scaffold, the 3-(4-hydroxyphenyl)indoline-2-ones. With humble beginnings in 19th century Bavaria, we review this scaffold's synthetic history and anticancer optimization, including its recent demonstration of tumor eradication of drug-resistant, estrogen receptor-positive breast cancer. Compounds containing the 3-(4-hydroxyphenyl)indoline-2-one pharmacophore are emerging as intriguing candidates for the treatment of cancer.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The University of Illinois has filed patents on some compounds described herein on which M. W. B. and P. J. H. are inventors. This intellectual property has been licensed to Systems Oncology, LLC, and P. J. H. is a consultant and a member of the Scientific Advisory Board of Systems Oncology.
Figures
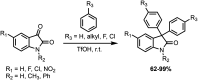
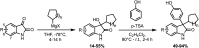



Similar articles
-
Design of a Structurally Novel Multipotent Drug Candidate by the Scaffold Architecture Technique for ACE-II, NSP15, and Mpro Protein Inhibition: Identification and Isolation of a Natural Product to Prevent the Severity of Future Variants of Covid 19 and a Colorectal Anticancer Drug.ACS Omega. 2022 Sep 10;7(37):33408-33422. doi: 10.1021/acsomega.2c04051. eCollection 2022 Sep 20. ACS Omega. 2022. PMID: 36157758 Free PMC article.
-
QSAR-based rational discovery of novel substituted-4'-iminospiro[indoline-3,3'-[1,2,5]thiadiazolidinyl]-2-one 1',1'-dioxide with potent in vitro anticancer activity.BMC Chem. 2019 Jan 29;13(1):3. doi: 10.1186/s13065-019-0520-z. eCollection 2019 Dec. BMC Chem. 2019. PMID: 31355364 Free PMC article.
-
Structure-activity study in the class of 6-(3'-hydroxyphenyl)naphthalenes leading to an optimization of a pharmacophore model for 17beta-hydroxysteroid dehydrogenase type 1 (17beta-HSD1) inhibitors.Mol Cell Endocrinol. 2009 Mar 25;301(1-2):205-11. doi: 10.1016/j.mce.2008.09.024. Epub 2008 Oct 4. Mol Cell Endocrinol. 2009. PMID: 18950679
-
Total Synthesis of Stelletins through an Unconventional Annulation Strategy.Acc Chem Res. 2021 Apr 6;54(7):1597-1609. doi: 10.1021/acs.accounts.0c00840. Epub 2021 Feb 26. Acc Chem Res. 2021. PMID: 33635622 Review.
-
Synthetic and Medicinal Perspective of Fused-Thiazoles as Anticancer Agents.Anticancer Agents Med Chem. 2021;21(11):1379-1402. doi: 10.2174/1871520620666200728133017. Anticancer Agents Med Chem. 2021. PMID: 32723259 Review.
Cited by
-
Single Dose of a Small Molecule Leads to Complete Regressions of Large Breast Tumors in Mice.ACS Cent Sci. 2025 Jan 22;11(2):228-238. doi: 10.1021/acscentsci.4c01628. eCollection 2025 Feb 26. ACS Cent Sci. 2025. PMID: 40028352 Free PMC article.
-
Identification of 3-((4-Hydroxyphenyl)amino)propanoic Acid Derivatives as Anticancer Candidates with Promising Antioxidant Properties.Molecules. 2024 Jun 30;29(13):3125. doi: 10.3390/molecules29133125. Molecules. 2024. PMID: 38999077 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

