Non-uniform distribution of dendritic nonlinearities differentially engages thalamostriatal and corticostriatal inputs onto cholinergic interneurons
- PMID: 35815934
- PMCID: PMC9302969
- DOI: 10.7554/eLife.76039
Non-uniform distribution of dendritic nonlinearities differentially engages thalamostriatal and corticostriatal inputs onto cholinergic interneurons
Abstract
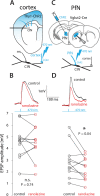
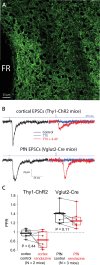
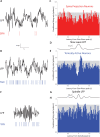
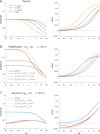
The tonic activity of striatal cholinergic interneurons (CINs) is modified differentially by their afferent inputs. Although their unitary synaptic currents are identical, in most CINs cortical inputs onto distal dendrites only weakly entrain them, whereas proximal thalamic inputs trigger abrupt pauses in discharge in response to salient external stimuli. To test whether the dendritic expression of the active conductances that drive autonomous discharge contribute to the CINs' capacity to dissociate cortical from thalamic inputs, we used an optogenetics-based method to quantify dendritic excitability in mouse CINs. We found that the persistent sodium (NaP) current gave rise to dendritic boosting, and that the hyperpolarization-activated cyclic nucleotide-gated (HCN) current gave rise to a subhertz membrane resonance. This resonance may underlie our novel finding of an association between CIN pauses and internally-generated slow wave events in sleeping non-human primates. Moreover, our method indicated that dendritic NaP and HCN currents were preferentially expressed in proximal dendrites. We validated the non-uniform distribution of NaP currents: pharmacologically; with two-photon imaging of dendritic back-propagating action potentials; and by demonstrating boosting of thalamic, but not cortical, inputs by NaP currents. Thus, the localization of active dendritic conductances in CIN dendrites mirrors the spatial distribution of afferent terminals and may promote their differential responses to thalamic vs. cortical inputs.
Keywords: basal ganglia; mouse; neuroscience; quasi-linear approximation; vervet.
© 2022, Oz et al.
Conflict of interest statement
OO, LM, AM, AK, NB, LT, HB, JG No competing interests declared
Figures


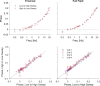
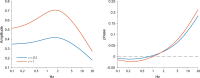
Similar articles
-
Parvalbumin-producing striatal interneurons receive excitatory inputs onto proximal dendrites from the motor thalamus in male mice.J Neurosci Res. 2018 Jul;96(7):1186-1207. doi: 10.1002/jnr.24214. Epub 2018 Jan 4. J Neurosci Res. 2018. PMID: 29314192
-
Cholinergic Transmission at Muscarinic Synapses in the Striatum Is Driven Equally by Cortical and Thalamic Inputs.Cell Rep. 2019 Jul 23;28(4):1003-1014.e3. doi: 10.1016/j.celrep.2019.06.077. Cell Rep. 2019. PMID: 31340139 Free PMC article.
-
A tonic nicotinic brake controls spike timing in striatal spiny projection neurons.Elife. 2022 May 17;11:e75829. doi: 10.7554/eLife.75829. Elife. 2022. PMID: 35579422 Free PMC article.
-
Excitatory extrinsic afferents to striatal interneurons and interactions with striatal microcircuitry.Eur J Neurosci. 2019 Mar;49(5):593-603. doi: 10.1111/ejn.13881. Epub 2018 Mar 25. Eur J Neurosci. 2019. PMID: 29480942 Free PMC article. Review.
-
Roles of centromedian parafascicular nuclei of thalamus and cholinergic interneurons in the dorsal striatum in associative learning of environmental events.J Neural Transm (Vienna). 2018 Mar;125(3):501-513. doi: 10.1007/s00702-017-1713-z. Epub 2017 Mar 21. J Neural Transm (Vienna). 2018. PMID: 28324169 Free PMC article. Review.
Cited by
-
Cholinergic control of striatal GABAergic microcircuits.Cell Rep. 2022 Oct 25;41(4):111531. doi: 10.1016/j.celrep.2022.111531. Cell Rep. 2022. PMID: 36288709 Free PMC article.
-
The Integration of Top-down and Bottom-up Inputs to the Striatal Cholinergic Interneurons.Curr Neuropharmacol. 2024;22(9):1566-1575. doi: 10.2174/1570159X22666231115151403. Curr Neuropharmacol. 2024. PMID: 38420787 Free PMC article.
-
Optogenetic Interrogation of Electrophysiological Dendritic Properties and Their Effect on Pacemaking Neurons from Acute Rodent Brain Slices.Bio Protoc. 2024 May 20;14(10):e4992. doi: 10.21769/BioProtoc.4992. eCollection 2024 May 20. Bio Protoc. 2024. PMID: 38798977 Free PMC article.
-
Distinct Modulation of I h by Synaptic Potentiation in Excitatory and Inhibitory Neurons.eNeuro. 2024 Nov 13;11(11):ENEURO.0185-24.2024. doi: 10.1523/ENEURO.0185-24.2024. Print 2024 Nov. eNeuro. 2024. PMID: 39406481 Free PMC article.
References
-
- Aosaki T, Tsubokawa H, Ishida A, Watanabe K, Graybiel AM, Kimura M. Responses of tonically active neurons in the primate’s striatum undergo systematic changes during behavioral sensorimotor conditioning. The Journal of Neuroscience. 1994;14:3969–3984. doi: 10.1523/JNEUROSCI.14-06-03969.1994. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

