Cost-Effectiveness of a Telephone-Based Smoking Cessation Randomized Trial in the Lung Cancer Screening Setting
- PMID: 35818125
- PMCID: PMC9382714
- DOI: 10.1093/jncics/pkac048
Cost-Effectiveness of a Telephone-Based Smoking Cessation Randomized Trial in the Lung Cancer Screening Setting
Abstract
Background: There are limited data on the cost-effectiveness of smoking cessation interventions in lung cancer screening settings. We conducted an economic analysis embedded in a national randomized trial of 2 telephone counseling cessation interventions.
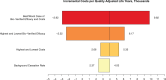
Methods: We used a societal perspective to compare the short-term cost per 6-month bio-verified quit and long-term cost-effectiveness of the interventions. Trial data were used to micro-cost intervention delivery, and the data were extended to a lifetime horizon using an established Cancer Intervention Surveillance and Modeling Network lung cancer model. We modeled the impact of screening accompanied by 8 weeks vs 3 weeks of telephone counseling (plus nicotine replacement) vs screening alone based on 2021 screening eligibility. Lifetime downstream costs (2021 dollars) and effects (life-years gained, quality-adjusted life-years [QALYs]) saved were discounted at 3%. Sensitivity analyses tested the effects of varying quit rates and costs; all analyses assumed nonrelapse after quitting.
Results: The costs for delivery of the 8-week vs 3-week protocol were $380.23 vs $144.93 per person, and quit rates were 7.14% vs 5.96%, respectively. The least costly strategy was a 3-week counseling approach. An 8-week (vs 3-week) counseling approach increased costs but gained QALYs for an incremental cost-effectiveness ratio of $4029 per QALY. Screening alone cost more and saved fewer QALYs than either counseling strategy. Conclusions were robust in sensitivity analyses.
Conclusions: Telephone-based cessation interventions with nicotine replacement are considered cost-effective in the lung screening setting. Integrating smoking cessation interventions with lung screening programs has the potential to maximize long-term health benefits at reasonable costs.
© The Author(s) 2022. Published by Oxford University Press.
Figures
Similar articles
-
A Randomized Trial of Telephone-Based Smoking Cessation Treatment in the Lung Cancer Screening Setting.J Natl Cancer Inst. 2022 Oct 6;114(10):1410-1419. doi: 10.1093/jnci/djac127. J Natl Cancer Inst. 2022. PMID: 35818122 Free PMC article. Clinical Trial.
-
Clinical impact and cost-effectiveness of integrating smoking cessation into lung cancer screening: a microsimulation model.CMAJ Open. 2020 Sep 22;8(3):E585-E592. doi: 10.9778/cmajo.20190134. Print 2020 Jul-Sep. CMAJ Open. 2020. PMID: 32963023 Free PMC article.
-
The Economic Impact of Smoking and of Reducing Smoking Prevalence: Review of Evidence.Tob Use Insights. 2015 Jul 14;8:1-35. doi: 10.4137/TUI.S15628. eCollection 2015. Tob Use Insights. 2015. PMID: 26242225 Free PMC article. Review.
-
Study protocol for a telephone-based smoking cessation randomized controlled trial in the lung cancer screening setting: The lung screening, tobacco, and health trial.Contemp Clin Trials. 2019 Jul;82:25-35. doi: 10.1016/j.cct.2019.05.006. Epub 2019 May 23. Contemp Clin Trials. 2019. PMID: 31129371 Free PMC article.
-
Varenicline: a pharmacoeconomic review of its use as an aid to smoking cessation.Pharmacoeconomics. 2010;28(3):231-54. doi: 10.2165/11204380-000000000-00000. Pharmacoeconomics. 2010. PMID: 20108995 Review.
Cited by
-
Effect of a mindfulness training app on a cigarette quit attempt: an investigator-blinded, 58-county randomized controlled trial.JNCI Cancer Spectr. 2023 Oct 31;7(6):pkad095. doi: 10.1093/jncics/pkad095. JNCI Cancer Spectr. 2023. PMID: 37951593 Free PMC article. Clinical Trial.
-
A Randomized Trial of Telephone-Based Smoking Cessation Treatment in the Lung Cancer Screening Setting.J Natl Cancer Inst. 2022 Oct 6;114(10):1410-1419. doi: 10.1093/jnci/djac127. J Natl Cancer Inst. 2022. PMID: 35818122 Free PMC article. Clinical Trial.
-
Understanding the perceived benefits, barriers, and cues to action for lung cancer screening among Latinos: A qualitative study.Front Oncol. 2024 Mar 20;14:1365739. doi: 10.3389/fonc.2024.1365739. eCollection 2024. Front Oncol. 2024. PMID: 38571494 Free PMC article.
-
Using the Past to Understand the Future of U.S. and Global Smoking Disparities: A Birth Cohort Perspective.Am J Prev Med. 2023 Apr;64(4 Suppl 1):S1-S10. doi: 10.1016/j.amepre.2022.12.003. Epub 2023 Feb 11. Am J Prev Med. 2023. PMID: 36781373 Free PMC article.
-
Strategies to deliver smoking cessation interventions during targeted lung health screening - a systematic review and meta-analysis.Chron Respir Dis. 2023 Jan-Dec;20:14799731231183446. doi: 10.1177/14799731231183446. Chron Respir Dis. 2023. PMID: 37311772 Free PMC article.
References
-
- American Cancer Society. Cancer facts and figures. 2022. https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts.... Accessed June 27, 2022.
-
- Moyer VA; U.S. Preventive Services Task Force. Screening for lung cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;160(5):330-338. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
