Copy number variation at the 22q11.2 locus influences prevalence, severity, and psychiatric impact of sleep disturbance
- PMID: 35820809
- PMCID: PMC9275284
- DOI: 10.1186/s11689-022-09450-0
Copy number variation at the 22q11.2 locus influences prevalence, severity, and psychiatric impact of sleep disturbance
Abstract
Background: Sleep disturbance is common, impairing, and may affect symptomatology in developmental neuropsychiatric disorders. Here, we take a genetics-first approach to study the complex role of sleep in psychopathology. Specifically, we examine severity of sleep disturbance in individuals with a reciprocal copy number variant (CNV) at the 22q11.2 locus and determine sleep's effect on psychiatric symptoms. CNVs (deletion or duplication) at this locus confer some of the greatest known risks of neuropsychiatric disorders; recent studies suggest the 22q11.2 deletion negatively impacts sleep, but sleep disruption associated with 22q11.2 duplication has not been investigated.
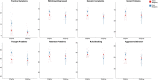
Methods: We compared subjective sleep disturbance and its relationship to psychiatric symptoms cross-sectionally and longitudinally over 1 year in 107 22q11.2 deletion (22qDel) carriers (14.56±8.0 years; 50% male), 42 22q11.2 duplication (22qDup) carriers (16.26±13.1 years; 54.8% male), and 88 age- and sex-matched controls (14.65±7.4 years; 47.1% male). Linear mixed models were used to compare sleep disturbance, assessed via the Structured Interview for Psychosis-Risk Syndromes (SIPS), across groups. Next, CNV carriers were categorized as good or poor sleepers to investigate sleep effects on multiple neurobehavioral traits: psychosis-risk symptoms (SIPS), autism-related behaviors (Repetitive Behavior Scale (RBS) and Social Responsiveness Scale (SRS)), real-world executive function (Behavior Rating Inventory of Executive Function (BRIEF)), and emotional/behavioral problems (Child Behavior Checklist (CBCL)). Linear mixed models tested the effect of sleep category and a group-by-sleep interaction on each measure, cross-sectionally and longitudinally.
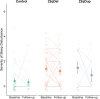
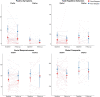
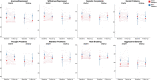
Results: 22qDel and 22qDup carriers both reported poorer sleep than controls, but did not differ from each other. Cross-sectionally and longitudinally, poor sleepers scored higher on positive symptoms, anxious/depressed, somatic complaints, thought problems, and aggressive behavior, as well as RBS and SRS total scores. There were significant group-by-sleep interactions for positive symptoms and the majority of CBCL subdomains, in which the difference between good and poor sleepers was larger in 22qDel compared to 22qDup.
Conclusions: Our findings indicate that CNVs at the 22q11.2 locus impact sleep which, in turn, influences psychopathology. Sleep disturbances can differentially impact psychopathology, depending on 22q11.2 gene dosage. Our findings serve as a starting point for exploring a genetic basis for sleep disturbance in developmental neuropsychiatric disorders.
Keywords: 22q11.2 locus; Autism spectrum disorder; Behavioral problems; Copy number variation; Developmental neuropsychiatric disorders; Executive function; Psychosis; Psychosis-risk symptoms; Schizophrenia; Sleep.
© 2022. The Author(s).
Conflict of interest statement
The authors declare they do not have competing interests.
Figures
Similar articles
-
Sleep in 22q11.2 Deletion Syndrome: Current Findings, Challenges, and Future Directions.Curr Psychiatry Rep. 2023 Oct;25(10):479-491. doi: 10.1007/s11920-023-01444-6. Epub 2023 Sep 18. Curr Psychiatry Rep. 2023. PMID: 37721640 Free PMC article. Review.
-
Distinct Neurocognitive Profiles and Clinical Phenotypes Associated with Copy Number Variation at the 22q11.2 Locus.medRxiv [Preprint]. 2023 May 16:2023.05.12.23289905. doi: 10.1101/2023.05.12.23289905. medRxiv. 2023. Update in: Autism Res. 2023 Dec;16(12):2247-2262. doi: 10.1002/aur.3049. PMID: 37292882 Free PMC article. Updated. Preprint.
-
Differential inflammatory profiles in carriers of reciprocal 22q11.2 copy number variants.Psychoneuroendocrinology. 2024 Nov;169:107135. doi: 10.1016/j.psyneuen.2024.107135. Epub 2024 Jul 20. Psychoneuroendocrinology. 2024. PMID: 39116521
-
Distinct neurocognitive profiles and clinical phenotypes associated with copy number variation at the 22q11.2 locus.Autism Res. 2023 Dec;16(12):2247-2262. doi: 10.1002/aur.3049. Epub 2023 Nov 23. Autism Res. 2023. PMID: 37997544 Free PMC article.
-
[An attempt to identify 22q11.2 microdeletions in samples of the Hungarian schizophrenia DNA bank by multiplex ligation-based probe amplification (MLPA): literature review, methodology and results].Neuropsychopharmacol Hung. 2016 Dec;18(4):209-218. Neuropsychopharmacol Hung. 2016. PMID: 28259864 Review. Hungarian.
Cited by
-
Sleep in 22q11.2 Deletion Syndrome: Current Findings, Challenges, and Future Directions.Curr Psychiatry Rep. 2023 Oct;25(10):479-491. doi: 10.1007/s11920-023-01444-6. Epub 2023 Sep 18. Curr Psychiatry Rep. 2023. PMID: 37721640 Free PMC article. Review.
-
Investigating the relationship between sleep disturbances and psychopathology In children and adolescents with microdeletion of 22q11 chromosome: an exploratory study.Front Psychiatry. 2025 Jul 23;16:1595492. doi: 10.3389/fpsyt.2025.1595492. eCollection 2025. Front Psychiatry. 2025. PMID: 40771649 Free PMC article.
-
Distinct Neurocognitive Profiles and Clinical Phenotypes Associated with Copy Number Variation at the 22q11.2 Locus.medRxiv [Preprint]. 2023 May 16:2023.05.12.23289905. doi: 10.1101/2023.05.12.23289905. medRxiv. 2023. Update in: Autism Res. 2023 Dec;16(12):2247-2262. doi: 10.1002/aur.3049. PMID: 37292882 Free PMC article. Updated. Preprint.
-
Sleep profiles in children with 22q deletion syndrome: a study of 100 consecutive children seen in a multidisciplinary clinic.J Clin Sleep Med. 2023 Jan 1;19(1):27-34. doi: 10.5664/jcsm.10238. J Clin Sleep Med. 2023. PMID: 35975550 Free PMC article.
-
Neurobehavioral Dimensions of Prader Willi Syndrome: Relationships Between Sleep and Psychosis-Risk Symptoms.Front Psychiatry. 2022 Apr 13;13:868536. doi: 10.3389/fpsyt.2022.868536. eCollection 2022. Front Psychiatry. 2022. PMID: 35492689 Free PMC article.
References
-
- Hertenstein E, Feige B, Gmeiner T, Kienzler C, Spiegelhalder K, Johann A, et al. Insomnia as a predictor of mental disorders: a systematic review and meta-analysis. Sleep Med Rev. 2019;43:96–105. - PubMed

